Pterygium is easy to describe in a simple way, but harder to model properly. It shows up as a tissue growth on the ocular surface, yet the disease involves much more than excess tissue alone. Inflammatory cells, vascular changes, stromal remodeling, and epithelial behavior all seem to be part of the picture. That is one reason pterygium eye models often fall short. They may capture one piece of the biology, but not the mix of cell types and signals that shape the disease over time.
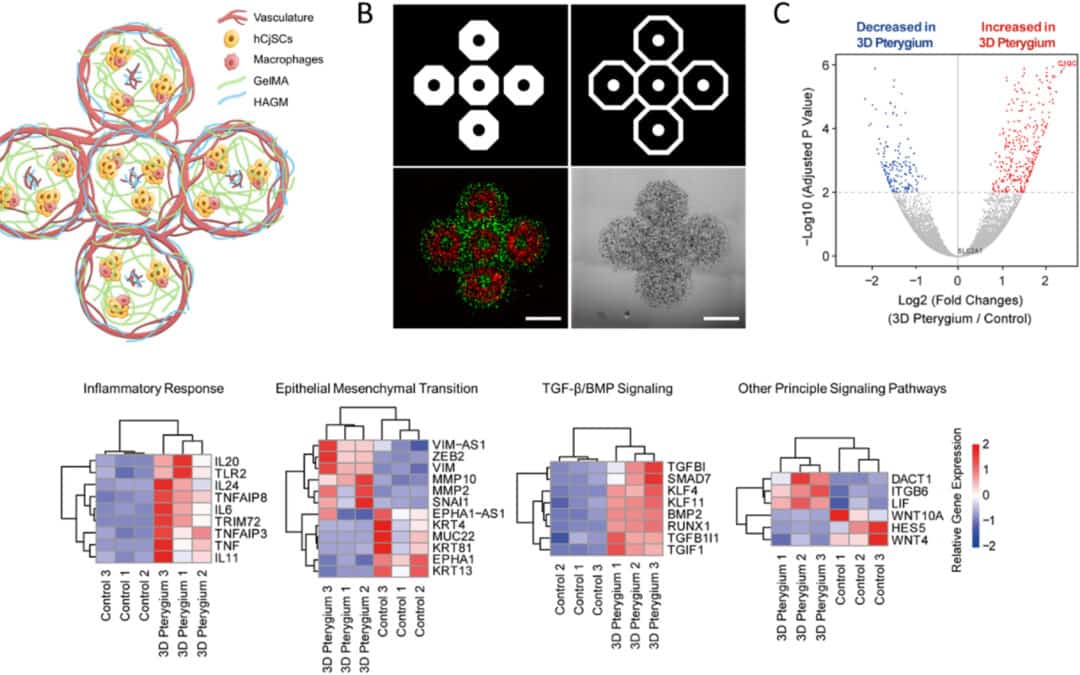
In this Biomaterials study out of UCLA and UC San Diego, the researchers moved away from the usual simpler setup and tried to build something closer to the tissue environment seen in pterygium. They used DLP 3D bioprinting to combine conjunctival stem cells with macrophages, endothelial cells, and stromal fibroblast-like cells in one construct. That gave them a pterygium eye model with more of the cell-to-cell complexity that is missing from standard cultures. They then checked the RNA profile and found that the printed model tracked with patient pterygium datasets in several of the pathways you would expect, especially inflammation, angiogenesis, and epithelial-mesenchymal transition.
Why a better pterygium eye model matters
Pterygium may seem straightforward on the surface, but it can be difficult to manage well. It can change the shape of the ocular surface, interfere with vision, and in some cases come back after surgery. Clinically, that makes it more than a minor irritation. From a research side, it is also a hard disease to model cleanly, which is part of why progress has been slower than people would like.
The authors describe pterygium as a vascularized conjunctival overgrowth linked to chronic inflammation and neovascularization, with conjunctival stem cells also likely involved. Existing models have tended to be either too simple or too inconsistent. Some lack the right mix of cell populations. Others are hard to scale or do not reproduce the disease environment well enough for useful screening. The aim in this paper was to build something more representative, a model that could be used not just to observe cells in culture, but to study disease mechanisms and test potential therapies in a more realistic setting.
How the model was built
The researchers first expanded primary human conjunctival stem cells using a feeder-free culture system. They then used a custom DLP-based 3D bioprinter to fabricate hydrogel scaffolds and later build a multicellular disease model.
The final printed pterygium construct had two layers:
-
a stem cell and macrophage layer to represent inflammatory cell infiltration
-
a vascular layer containing endothelial cells and fibroblast-like support cells to represent angiogenesis
This layered design is why the pterygium eye model is more than a simple monoculture or a basic hydrogel cell encapsulation study. It was meant to recreate the disease microenvironment in a structured way.
Mechanical characterization of the printed layers
Mechanical characterization of the printed layers helps ensure the model replicates the stiffness-dependent behaviour of native ocular tissue and supports the right cell state during disease modeling.
Before building the disease model, the team mechanically tested the printed hydrogel conditions using a CellScale MicroTester. They printed small GelMA cylinders and measured compressive Young’s modulus to compare a softer and stiffer scaffold formulation. The soft condition was about 2.98 ± 0.85 kPa, while the stiff condition was about 11.20 ± 0.62 kPa.
That detail matters because the mechanical properties influenced the biology.
Why micro-mechanical testing mattered
This study is a good example of why micro-mechanical testing belongs in bioprinting workflows. The team was not just printing a structure that looked right. They were testing whether the printed environment had the right physical properties to support conjunctival stem cells.
Because the constructs were small and soft, micro-mechanical testing was the right scale. In this case, compression testing of soft hydrogel tissues using the MicroTester helped determine which scaffold stiffness better preserved cell viability and function. That is an important point for the new site because it connects the printed disease model directly to mechanical testing, rather than treating mechanics as background information.
Bioink and 3D bioprinting materials used in the model
The printed structures were based on GelMA and HAGM hydrogels, with different formulations used depending on the cell population and layer. The researchers note that GelMA was chosen because of its strong cell-binding capacity and prior success in stem cell encapsulation. For the conjunctival stem cell printing, they compared soft and stiff GelMA-only conditions, then used the softer formulation moving forward. A different GelMA-HAGM formulation was used for the vascular cell printing.
What the transcriptomic analysis showed
Once the multicellular model was built, the researchers used RNA sequencing to compare the 3D pterygium model with controls. The analysis showed major transcriptomic differences, including upregulation of genes linked to:
-
inflammatory response
-
epithelial-mesenchymal transition
-
TGF-β/BMP signaling
-
TNF-α/NF-κB signaling
-
angiogenesis-related pathways
The model also aligned with published RNA-seq datasets from patient-derived pterygium tissue, which is one of the strongest parts of the paper. The 3D model was not just biologically active. It showed molecular features consistent with actual disease.
Why this matters for pterygium research
The value of this work is that it moves pterygium eye models closer to something useful for both mechanism studies and therapy development. The model combines stem cells, inflammation, and vascular components in a way that more closely resembles the real ocular surface environment than standard monoculture systems.
That makes it relevant for:
-
organoid and tissue mimetic systems
-
bioink and 3D bioprinting materials
-
ocular disease modeling
-
drug testing and personalized medicine
-
micro-mechanical testing of soft printed tissues
Conclusion
Using DLP-based 3D bioprinting, the researchers built a multicellular system that included conjunctival stem cells, immune cells, and vascular cells to mimic the pterygium microenvironment. They also did not treat the scaffold as just a neutral container for the cells. The group checked its stiffness with MicroTester compression testing first, then used that information to choose the softer hydrogel, since that version gave better viability and better retention of the stem-cell-related markers.
That is really what gives the study more value. The printed construct was shaped by the mechanics as well as the biology, and the final pterygium eye model was backed up by transcriptomic data rather than appearance alone. It is not only a printed eye model. It is a mechanically informed disease model that could support future pterygium research, drug testing, and personalized ophthalmic applications.
Read the full study here.