Researchers at Auburn University developed a prostate cancer tissue model by starting with a simple but important question: how stiff is the tumour tissue they are actually trying to mimic? Instead of choosing an arbitrary material formulation, they measured the mechanical properties of PC-3 xenograft tumours first and then used those measurements to guide the design of their engineered tissues. That gives the model a stronger biological starting point than many in vitro cancer systems.
The result was a tunable 3D co-culture system built from prostate cancer cells and fibroblasts, designed to reproduce the heterogeneous mechanical environment of real tumours. That matters because tumour stiffness is not just a background property. In prostate cancer, it is linked to changes in gene and protein expression, therapeutic response, and aggressive disease behaviour.
Why tumour stiffness matters in a prostate cancer tissue model
A useful prostate cancer tissue model should do more than keep cells alive in 3D. It should also expose those cells to biomechanical cues that are close to what they would experience in a tumour. In this study, the authors make that point clearly: if an in vitro model does not capture pathophysiologically relevant stiffness, it may miss tumour behaviours that matter for progression and drug response.
That is part of what makes this work notable. The model was not only built as a 3D scaffold. It was built as a mechanically informed platform for studying the tumour microenvironment.
For a broader introduction to how soft tissues and biomaterials are mechanically characterized, see our guide to mechanical testing of biomaterials.
How xenograft tumour stiffness was measured
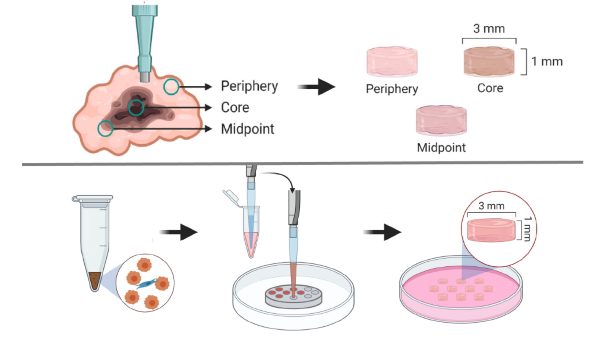
The team generated PC-3 prostate tumour xenografts in mice, then excised the tumours, sliced them into roughly 1 mm sections, and used a 3 mm biopsy punch to collect specimens from the geometric core, midpoint, and periphery. Those samples were then mechanically tested right away. Figure 1 in the publication shows that workflow clearly, including the regional sampling strategy used to capture heterogeneity within the tumour mass.
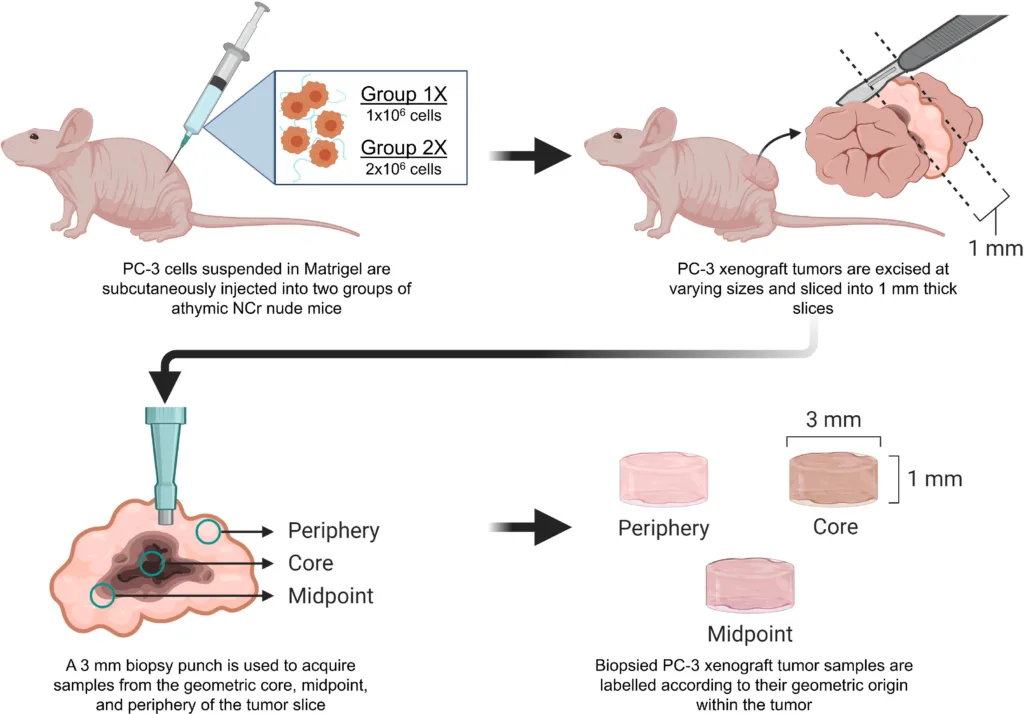
Adapated from Habbit et al. Tunable three-dimensional engineered prostate cancer tissues for in vitro recapitulation of heterogeneous in vivo prostate tumor stiffness. Acta Biomaterialia. 2022.
For the stiffness measurements, the tumour samples were analysed by parallel plate compression under physiological conditions using a MicroTester (AKA ‘MicroSquisher’ in the paper) apparatus from CellScale. The paper reports that Group 1X tumours ranged from about 95 to 3,500 Pa overall, while Group 2X tumours ranged from about 230 to 6,750 Pa, with higher cell seeding density producing broader and stiffer tumour ranges.
What the xenograft results showed
One of the most useful findings is that xenograft tumor stiffness was not uniform. Stiffness changed both across different tumours and within different parts of the same tumour. In the Group 2X samples, the outer regions generally measured stiffer than the core, which is a good example of why one average value cannot fully capture the mechanical landscape of a tumour.
The authors also showed that changing the initial cancer cell number changed the resulting tumour mechanics. When the seeding density was doubled, the measured stiffness range became broader and extended to higher values. That matters for modelling because it suggests tumour stiffness is influenced by how the tissue grows and organizes over time, not just by its final dimensions.
How the engineered prostate cancer tissue model was built
The in vitro system was a co-culture of PC-3 prostate cancer cells and BJ-5ta fibroblasts encapsulated within a PEG-fibrinogen matrix. The researchers then modulated the matrix using added PEGDA so they could tune stiffness across a wide range. That allowed them to create engineered prostate cancer tissues, or EPCaTs, with matrix conditions that overlapped the measured xenograft values.
This is one of the strongest parts of the paper. The prostate cancer tissue model was not built around a single “best” stiffness. It was designed as a tunable system, which is much more realistic for tumour biology.
Micro-mechanical testing linked the in vivo and in vitro models
In practical terms, compressive micro-mechanical testing was used to quantify stiffness differences between in vivo xenograft tumours and engineered tissue constructs. The paper describes parallel plate compression performed in a fluid bath at 37 °C to generate force-displacement data and calculate Young’s modulus for both in vivo and in vitro tissues.
The engineered tissues could match the xenograft stiffness range
Across different matrix formulations and culture times, the EPCaT model achieved a stiffness range of about 50 to 10,000 Pa. That span was broad enough to cover the stiffness measured from the xenograft samples and even extend into published clinical prostate tissue values discussed by the authors.
The strongest overlap was also biologically useful. Softer PF and PF+1%PEGDA tissues aligned more closely with the softer Group 1X xenografts, while PF+2%PEGDA and PF+3%PEGDA tissues aligned more closely with the stiffer Group 2X tumours. That means the prostate cancer tissue model can be adjusted depending on which tumour-like mechanical environment a researcher wants to study.
Why stiffness matching matters
Stiffness matching matters because cells respond to mechanics. In this paper, increased matrix stiffness was associated with enrichment of gene sets linked to tumourigenic progression. The model therefore gives researchers a way to study not just cancer cells in 3D, but cancer cells in a mechanically relevant environment that may influence signalling, phenotype, and therapeutic response.
That is a big step for disease modelling and drug development. A cancer model that is easier to tune toward the mechanics of site-of-origin or soft-tissue metastatic disease may be more useful than a generic 3D culture that has the wrong physical context.
For a related example of how mechanics and stromal interactions shape tumour behaviour, read our article on tumor stiffness and cancer-associated fibroblasts.
What the MicroTester contributed
Researchers used the CellScale MicroTester to measure stiffness via micro-mechanical testing. That testing created the bridge between the xenograft tissue and the engineered tissue model. Without that mechanical comparison, the in vitro platform would not have been informed by real tumour properties in the same way.
Read the full publication here.
Related reading
If your work involves smaller engineered tissues or mechanically sensitive samples, you may also be interested in our article on micro-testing cell clusters.
You can also read more about other CellScale medical models in the brain dura mater here.