Developing a miniaturized heart valve is of course not a simple exercise. Even the process of shrinking existing devices isn’t really applicable. That’s because once valve diameters drop below 10 mm, many of the assumptions that hold for adult or even paediatric implants start to break down. Leaflet thickness becomes comparable to the vessel diameter. Sutures occupy a non‑negligible fraction of the structure. Small changes in material compliance begin to alter how the valve opens, closes, and survives repeated loading.
A recent peer‑reviewed study from researchers at RWTH Aachen University (and collaborators) explores this problem directly, focusing on a miniaturized heart valve intended for use in the ductus arteriosus of infants with hypoplastic left heart syndrome (HLHS). Rather than adapting an existing valve concept, the authors start from the constraints of the anatomy and work outward, using material selection and mechanical testing (with the CellScale UniVert) to guide each design decision.
Why miniaturized heart valves are different from scaled‑down valves
Most commercially available paediatric heart valve replacements have diameters of 12–16 mm. These work in many cases, but not for the ductus arteriosus (targeted in HLHS interventions). The ductus arteriosus typically measures only 6–8 mm. At this scale, simply scaling down leaflet geometries or stent designs introduces new mechanical and hydrodynamic problems.
In HLHS patients, the ductus arteriosus is kept open to support systemic circulation. Without a valve, some degree of backward flow through the ductus is unavoidable, particularly during diastole, and this can place additional load on the right heart. The approach explored in this study integrates a miniaturized heart valve directly into a ductus arteriosus stent, with the goal of limiting backflow during diastole rather than replicating a lifelong valve replacement.

Figure 1. Miniaturized heart valve concept for hypoplastic left heart syndrome (HLHS).
Schematic showing altered blood flow pathways in HLHS patients treated with a ductus arteriosus stent, and the integration of a miniaturized heart valve within the stent to limit backflow. By regulating flow through the ductus arteriosus, the valve aims to reduce systemic pressure on the right heart and improve flow balance between pulmonary and systemic circulation. Adapted from Sonntag et al., Bioactive Materials (2026).
A biohybrid approach to miniaturized heart valve materials
To meet the competing demands of flexibility, strength, and extreme miniaturization, the valve leaflets are fabricated as a biohybrid heart valve structure. The mechanical backbone consists of an electrospun thermoplastic polyurethane (TPU) scaffold, produced as a thin tubular construct with controlled wall thickness.
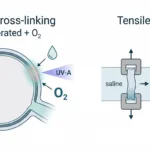
After electrospinning, the TPU scaffold is dip-coated with elastin-like recombinamers using a layer-by-layer process. The ELR solution penetrates the fibre network during coating and remains associated with the scaffold after crosslinking. Following this step, the tubular scaffold no longer permits fluid to pass through the wall under static conditions.
The coated tube is then assembled into a three-leaflet valve inside a stent. Leaflets are formed by suturing the tube at three commissural points, rather than by moulding or cutting the material.

Figure 2. Biohybrid fabrication and valve shaping for a miniaturized heart valve.
Schematic and microscopy images showing dip-coating of an electrospun TPU scaffold with elastin-like recombinamers, followed by shaping of the coated tube into a three-leaflet valve inside a stent. Cross-sections illustrate ELR infiltration and sealing of the fibrous scaffold, while the commissural attachment points define leaflet formation. Adapted from Sonntag et al., Bioactive Materials (2026).
Mechanical testing of a miniaturized heart valve scaffold
Mechanical testing plays a central role in this study, not simply as validation but as a design filter. Tensile testing, cyclic loading, and suture retention testing were completed with the CellScale UniVert, and used to assess how the scaffold behaves under deformation relevant to neonatal circulation.
Similar mechanical considerations have been explored in studies examining regional variations of native heart valve leaflets.
In this study, stress–strain curves show that the electrospun TPU exhibits large strain capacity, with elongation well beyond physiological requirements. Tensile force and peak tension increase with scaffold thickness across the tested range. After ELR coating, the electrospun scaffold no longer allows fluid to pass through the fibre network, and burst pressure can be measured without leakage through the wall.
Cyclic testing reveals time-dependent viscoelastic behaviour, with initial stress relaxation followed by stabilisation over repeated loading. Based on these measurements, subsequent testing focused on a narrower range of scaffold thicknesses.

Figure 3. Mechanical testing with the UniVert, used to define material limits in a miniaturized heart valve scaffold.
Uniaxial tensile and cyclic loading data for electrospun TPU scaffolds of different thicknesses, showing large strain capacity, thickness-dependent tension, and viscoelastic behaviour under repeated loading. ELR coating seals the fibrous scaffold, enabling burst pressures well above physiological ranges. These measurements informed leaflet thickness selection for the final valve design. Adapted from Sonntag et al., Bioactive Materials (2026).
Deployment and hydrodynamic behaviour at neonatal scale
Beyond material coupons, the valve must function once crimped, delivered, and deployed in a vessel that closely matches the ductus arteriosus. The assembled miniaturized heart valve is crimped to catheter-compatible diameters and delivered into a compliant vascular graft representative of neonatal anatomy.
After release from the delivery system, the stent expands within the graft and the valve leaflets return to their deployed configuration. The assembled valve was then evaluated in a pulsatile flow loop, where leaflet motion during opening and closing was recorded and flow and pressure were measured across the valve.

Figure 4. Deployment and hydrodynamic function of a miniaturized heart valve in a native-like vessel.
Images show crimping of the valve for catheter-based delivery, followed by self-expansion and placement within a compliant vascular graft. Hydrodynamic testing under pulsatile flow confirms stable valve function after deployment. Adapted from Sonntag et al., Bioactive Materials (2026).
Failure analysis and reinforcement in miniaturized cardiovascular devices
As with many miniaturized cardiovascular devices, failure modes shift as structures become thinner and more constrained. Fatigue testing reveals that early failure consistently occurs at the commissural suture points, where the leaflet material experiences local stress concentrations.
Rather than changing the base material, the authors introduce a targeted textile reinforcement at these locations. This simple modification increases suture retention force and extends fatigue life by several fold, without substantially altering leaflet thickness or compliance.

Figure 5. Failure analysis and reinforcement strategy for a miniaturized heart valve.
Fatigue testing identifies commissural suture points as the primary failure locations. Adding local textile reinforcement increases suture retention force and extends fatigue life, illustrating how mechanical testing can directly inform design iteration in miniaturized heart valves. Adapted from Sonntag et al., Bioactive Materials (2026).
What this study shows about designing at neonatal scale
One thing that stands out in this work is how quickly mechanics become a limiting factor once cardiovascular devices are reduced to neonatal dimensions. At this scale, leaflet thickness, suture placement, and local reinforcement are no longer secondary details. They begin to define whether a device can be fabricated reproducibly, survive cyclic loading, and function once deployed.
In this study, mechanical testing with the UniVert is carried out alongside material fabrication and assembly, rather than being confined to a single evaluation step at the end. Measurements of tensile behaviour, burst pressure, and fatigue response are reported across multiple scaffold configurations, with testing repeated after changes to thickness, coating, and reinforcement strategy. In practice, this shortens the distance between material characterisation and device iteration.
For miniaturized heart valve development, particularly in congenital heart disease applications where anatomy leaves little margin for error, this kind of mechanics-driven workflow is increasingly difficult to avoid. As devices continue to shrink, it becomes less useful to ask whether a material is “strong enough” in isolation. The more relevant question is how it behaves once assembled, constrained, sutured, crimped, and repeatedly loaded inside a vessel only a few millimetres wide.
Read the Full Publication
About the CellScale UniVert
The CellScale UniVert is used for uniaxial tensile, compression, cyclic, fatigue, and viscoelastic testing of biomaterials and soft tissues, with force capacities up to 200N. It is commonly applied in studies involving highly compliant or delicate specimens, including electrospun scaffolds, hydrogels, soft polymers, and engineered tissues.
In this study, the UniVert was used to perform uniaxial tensile testing, cyclic loading, and suture retention testing of electrospun TPU scaffolds under hydrated, temperature-controlled conditions. These measurements helped define acceptable thickness ranges, identify failure-prone regions, and guide reinforcement strategies for the miniaturized heart valve design.
Similar uniaxial testing approaches are used in other studies involving thin, compliant materials tested under physiological conditions.