Antifreeze protein delivery sounds a little unusual at first, but the problem behind it is fairly practical. Antifreeze proteins have been studied for years because of the way they interact with ice crystal growth and ice recrystallization, which makes them interesting for cryopreservation and, at least in principle, cold-injury prevention.
The trouble is getting them where they need to go. These are not small, easy molecules to diffuse across tissue barriers, and conventional oral, intramuscular, or intravenous routes do not necessarily solve the local delivery problem in skin or other targeted tissues.
That is the setup for a recent ACS Omega paper from researchers at Boise State University and collaborators. Instead of trying to force systemic delivery to do a local job, the group built silk fibroin microneedle patches designed for antifreeze protein delivery into the epidermis.
They also asked a fairly practical mechanics question early: are the patches robust enough to penetrate skin without the protein payload changing how they behave? To check that, the team used a CellScale UniVert for compression testing alongside the release and skin-delivery experiments, so the paper ends up being about more than patch fabrication alone.
One thing that stands out is that the paper keeps returning to a practical constraint. If the cargo is structurally sensitive, then the fabrication method matters just as much as the final patch geometry. The authors framed this around a heat-free process for silk microneedles, then followed that with release, activity, and skin-insertion data rather than stopping at microscopy images alone.
Why antifreeze protein delivery is difficult
The core issue in antifreeze protein delivery is not whether antifreeze proteins are functional. It is whether they can be delivered locally, at useful concentrations, without losing function along the way. The paper notes that their molecular size and charge limit cellular uptake and have made it difficult to target tissues effectively through more traditional routes. That matters even more if the intended use case is localized, whether that is skin-facing protection from cold injury or delivery to defined tissue regions in a preservation setting.
The authors position transdermal delivery as a way around that bottleneck. Microneedles can breach the stratum corneum and reach the epidermis without relying on passive diffusion through intact skin, and that makes them a logical platform for antifreeze protein delivery. In this study, the material choice was silk fibroin, which is already familiar in biomaterials work and can be processed in ways that help stabilize delicate cargo. The interesting part here is not silk on its own, but silk used in a fabrication workflow that avoids post-processing steps likely to damage a fragile protein therapeutic.
How were silk microneedles built for antifreeze protein delivery?
The microneedle patches were fabricated as 20 x 20 arrays in PDMS molds, giving 400 needles per patch. The target geometry came from a printed inverse mold with 700 μm needle height and 400 μm base width, while the final dried patches measured roughly 620 to 660 μm in height with an average base diameter of 450 μm. The authors note that this was still within about 10% of the mold dimensions, and that the dimensional shift did not appear to impair penetration into porcine skin.
The fabrication detail that matters most for antifreeze protein delivery is that the loaded silk solution was cast and dried without hot or cold processing steps during encapsulation. The patches were produced from 7% or 10% silk fibroin formulations, and the antifreeze protein cargo was concentrated into the needle region before the non-loaded backing layer was added. The study reports that stored patches showed no meaningful difference in performance or AFP activity whether they were kept at 25 °C, 4 °C, or -80 °C.

Morphology of the silk fibroin microneedle patch used for antifreeze protein delivery. Panel A shows the full patch, Panels B and C show the regular needle array and tip geometry at higher magnification, and Panels D and E compare unloaded needles with AFP-loaded needles, where the yellow-green appearance in E reflects tagged protein within the patch. Adapted from Penney BT, et al. Silk Microneedles for Targeted Epidermal Delivery of Antifreeze Proteins. ACS Omega. 2026.
Can silk microneedles deliver antifreeze proteins without damaging them?
This is where the paper starts to become more than a manufacturing exercise. The authors loaded AFP-III into the silk microneedles across a range of doses and then checked both release and retained function. In deionized water, about 50% of the AFP payload was released within 15 minutes, and about 70% was released by 400 minutes. They also report an average mass loss of 32.0 ± 1.0% from the patches, which is a useful reminder that these are dissolving delivery structures, not permanent implants.
More importantly, the released AFPs still showed thermal hysteresis activity around 0.3 °C, comparable to native AFP-III at similar concentrations. In practice, that is one of the more important checks in the whole study. A delivery platform for fragile biologics is not very interesting if the cargo comes out inactive. Here, the data suggest that antifreeze protein delivery through these silk microneedles preserved enough structure and function for the proteins to retain measurable antifreeze behaviour after release.
The authors also note that AFP incorporation did not visibly disrupt patch formation, needle morphology, or mechanical properties. That is helpful because it suggests the loaded version behaved much like the unloaded version from a structural standpoint, rather than turning into a softer or less reliable patch as soon as a protein payload was introduced.
Mechanical testing of silk microneedles for antifreeze protein delivery
The group did not just assume the microneedles would be stiff enough. They compression-tested quartered silk fibroin patches using a CellScale UniVert mechanical tester with a 5 kg load cell, a 0.1 N preload, and loading to roughly 35 to 40% strain at 1% strain/s. Modulus was taken from the linear portion of the force-displacement response.
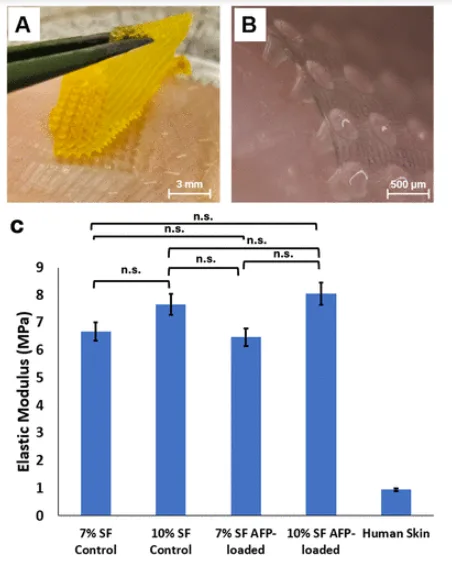
Mechanical testing and skin insertion data for the silk microneedle patch used in antifreeze protein delivery. Panel A shows a quartered AFP-loaded patch after compression into porcine skin, Panel B shows needle insertion into the outer epidermis, and Panel C compares elastic modulus across 7% and 10% silk formulations with and without AFP loading. The lack of significant stiffness change after loading is one of the more useful checks in the paper. Adapted from Penney BT, et al. Silk Microneedles for Targeted Epidermal Delivery of Antifreeze Proteins. ACS Omega. 2026.
The reported elastic moduli were 6.67 MPa for the 7% silk formulation and 7.55 MPa for the 10% silk formulation. Antifreeze protein loading did not significantly change stiffness, and the paper reports no significant difference between loaded and unloaded patches in either formulation. That is a fairly practical result. If the loaded patch had become mechanically unreliable, the whole antifreeze protein delivery concept would have become much harder to defend. Instead, the data suggest that adding AFPs did not meaningfully compromise compression behaviour.
The authors then paired those measurements with insertion observations in porcine skin. They report penetration depths of roughly 100 to 300 μm into the upper epidermal layer, with post-compression reductions in needle height of 40 to 100 μm and tip blunting or breakage in more than 75% of needles after compression. That last detail is worth noticing. The needles are not presented as perfectly unchanged after use. They are presented as strong enough to do the job and then dissolve, which is a more realistic framing for this kind of device.
If readers want more context on why soft biomaterial devices are not always straightforward to test mechanically, our post on mechanical testing of biomaterials is useful. There is also a nice parallel with our eariler highlight on smart bandage technology, where microstructured, skin-facing devices also had to be understood as mechanical systems rather than just material concepts.
Can microneedles deliver antifreeze proteins into skin?
This is really the question the paper has been working toward. Fabrication is one thing, and compression data are necessary, but antifreeze protein delivery only becomes convincing if the proteins can actually be shown inside tissue after application.
To test that, the authors used ex vivo porcine skin. That choice is not incidental. They make the case that porcine skin is a more appropriate analogue for human skin than the rodent models commonly used in the microneedle literature. The patches were inserted with a controlled clamp system, and fluorescently labelled AFP-loaded patches were applied for 30 to 60 minutes before the tissue was fixed, cryosectioned, and imaged by confocal microscopy.
What they observed is what makes the study feel worth writing about. The fluorescence signal was strong in the epidermis after application of AFP-loaded patches, and it was not confined only to the exact needle insertion points. The authors note that fluorescence extended deeper than the nominal needle position, suggesting diffusion beyond the initial insertion zone. They also show a control condition with minimal signal, which makes the delivery result easier to trust.
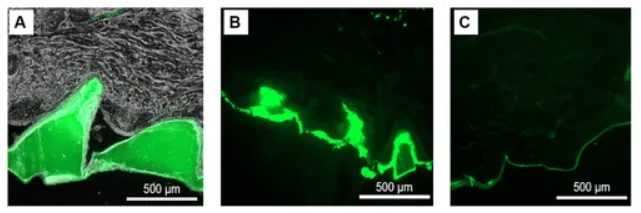
Evidence of antifreeze protein delivery into porcine skin. Panel A shows fluorescence confined to AFP-loaded microneedles immediately after insertion, Panel B shows signal throughout the epidermis after 30 minutes of application and patch removal, and Panel C shows the low-fluorescence control. Panel B is the key panel here because it shows release and dispersion beyond the original needle positions. Adapted from Penney BT, et al. Silk Microneedles for Targeted Epidermal Delivery of Antifreeze Proteins. ACS Omega. 2026.
There is still some uncertainty here, and the paper is fairly open about it. They did not directly quantify the exact amount of AFP delivered into the porcine skin, and they did not test whether that delivered protein actually reduced cold damage in tissue. So this is best read as proof-of-concept antifreeze protein delivery into a human-relevant skin analogue, not as a demonstration of frostbite prevention. Still, as an early step, it is a meaningful one.
What makes antifreeze protein delivery with silk microneedles interesting?
One reason this paper works well is that it sits at the intersection of biomaterials processing, transdermal drug delivery, and mechanical validation. It is not only about a new therapeutic idea, and it is not only about patch fabrication. It is about whether a structurally sensitive protein can be packaged into a dissolving microneedle system, survive processing, stay active after release, and reach the epidermis in a meaningful way.
There is also a broader biomaterials angle here. The authors argue that the heat-free silk fibroin process may be applicable to other peptides and proteins, not just antifreeze proteins. They specifically point to avoiding chemical cross-linkers and water-vapour or heat annealing, instead relying on silk crystallization during drying to achieve mechanical integrity. In practice, that may matter for anyone thinking about microneedle delivery of labile cargo where aggressive post-processing is likely to be a problem.
For readers interested in related skin-facing biomaterial work, our research highlight on oxygen-sensing 3D printed skin grafts is a recommended read. It is a different application, but it shares the same general theme that tissue-facing biomaterials often have to be understood through both structure and mechanics before the biological story becomes convincing.
Where could antifreeze protein delivery go next?
The paper leans toward cryopreservation and cold-injury applications, and that seems reasonable, though still preliminary. The authors suggest that local delivery of AFPs into skin could eventually be explored for frostbite prevention, and that targeted delivery into specific tissue regions might also be useful in preservation workflows for donor tissues or organs. At the same time, they are careful enough to note the study’s limitations: no in vivo testing yet, no direct assessment of cold protection in tissue, no exact quantification of delivered AFP in skin, and release testing done in deionized water rather than more physiologically relevant buffers.
That restraint is probably the right way to read the work. It appears to be an encouraging first demonstration of antifreeze protein delivery into porcine epidermis using silk microneedles, with functional retention of the protein cargo and enough mechanical robustness to support insertion. Whether that turns into a practical frostbite or organ-preservation technology is still an open question. But as a study, it does a good job of answering the first set of questions that needed to be answered.
Final thoughts on antifreeze protein delivery with silk microneedles
There are papers where the main contribution is a new material recipe, and there are papers where the interesting part is the biological outcome. This one sits somewhere in between. The silk fibroin microneedles matter because they were built around a delivery problem that has not been easy to solve through conventional routes. The antifreeze proteins matter because the authors checked whether they still worked after encapsulation and release. The mechanics matter because none of the delivery story means much unless the patch can actually enter tissue.
The use of compression testing on the UniVert fits naturally into that workflow. It is part of the paper’s answer to a practical question: if a dissolving microneedle patch is supposed to carry fragile cargo into skin, how stiff does it need to be, and does the loaded version still behave the way the unloaded one does? In this case, the answer appears to be yes, at least well enough for ex vivo porcine insertion and epidermal delivery.
About the UniVert
In this study, the CellScale UniVert was used to compression test the silk microneedle patches before skin insertion. That step helped the researchers check whether the patches were mechanically robust enough to penetrate porcine skin and whether loading them with antifreeze proteins changed their stiffness. The results suggested that AFP loading did not significantly alter patch mechanics, which made the delivery data easier to interpret.
More broadly, the UniVert is used for mechanical testing of small and soft samples where controlled loading and straightforward test setup matter, including biomaterials, hydrogels, soft tissues, scaffolds, and other natural and engineered constructs. In papers like this one, it becomes part of a fairly practical workflow: first confirm that the structure can withstand loading, then look at how it performs in the biological application.
Citation