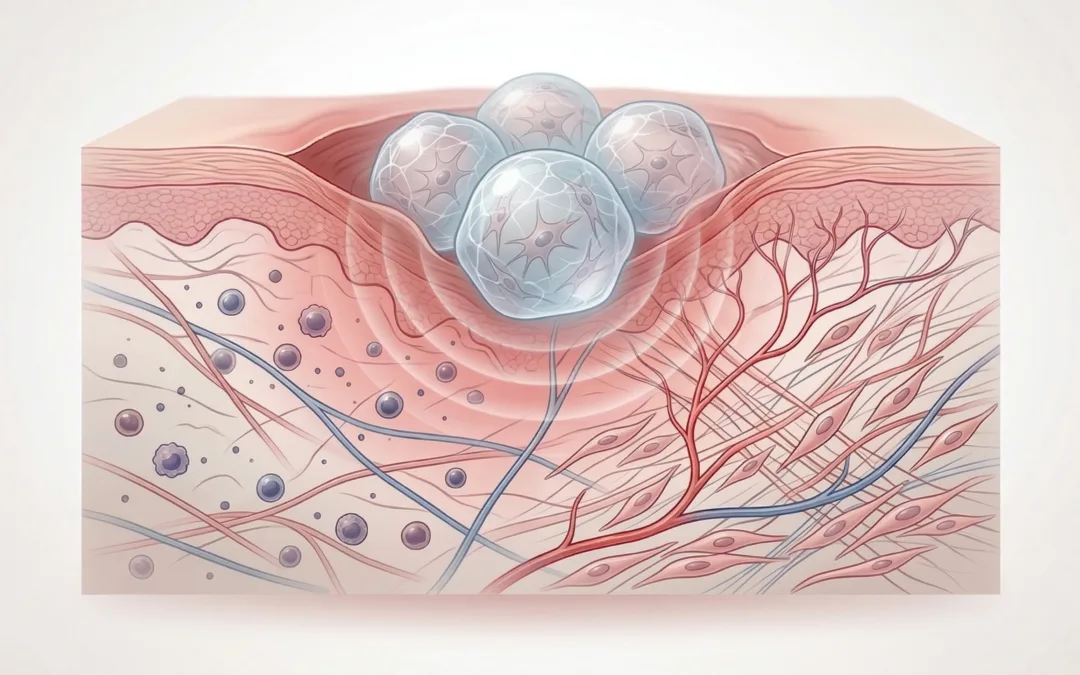
Chronic diabetic wounds are slow, but the deeper issue is that they stall. The inflammatory phase persists, angiogenesis stays weak, and remodeling does not kick in the way it should.
That is part of what makes this study out of Sun Yat-sen University in China interesting. The authors were not only asking whether mesenchymal stromal cells could help. They were asking whether the local mechanical environment around those cells could be tuned in a way that changes what the cells actually do once delivered.
Using gelatin methacryloyl (GelMA), the group built cell-laden hydrogel microspheres with two different stiffness states, then paired those mechanical conditions with inflammatory priming using TNF-α and IFN-γ. The idea was fairly direct: if diabetic wounds already present inflammatory cues, can a mechanically tuned carrier push MSCs toward a more useful secretory response for diabetic wound repair?
Mechanical characterization of the microspheres was performed using a CellScale MicroTester, which matters here because the whole argument depends on the soft and stiff groups being meaningfully different, not just nominally different.
What stands out in practice is that this is not really a story about putting cells into a gel and hoping for the best. It is more a story about treating matrix mechanics as an experimental input. The soft microspheres behaved differently, the encapsulated cells behaved differently, and later the wound model behaved differently too.
What are MSC hydrogel microspheres for diabetic wound healing trying to solve?
The paper starts with what clinicians and researchers see all the time in diabetic wounds: inflammation lingers, vascular support is limited, and closure processes like re-epithelialization and remodeling do not progress on schedule. MSC hydrogel microspheres for diabetic wound healing are appealing in that setting because MSCs can release paracrine signals tied to immune modulation, matrix remodeling, and angiogenic support. But the authors point out that MSC function depends heavily on context, and that low-grade chronic inflammation may not necessarily engage the most therapeutically useful MSC response on its own.
That is where the hydrogel microspheres come in. By encapsulating MSCs together with defined inflammatory factors inside a controllable GelMA microenvironment, the group tried to create localized chemomechanical cues rather than leaving the cells to interpret a messy wound environment without much structure. The microspheres were designed as injectable carriers, but also as small mechanical niches.
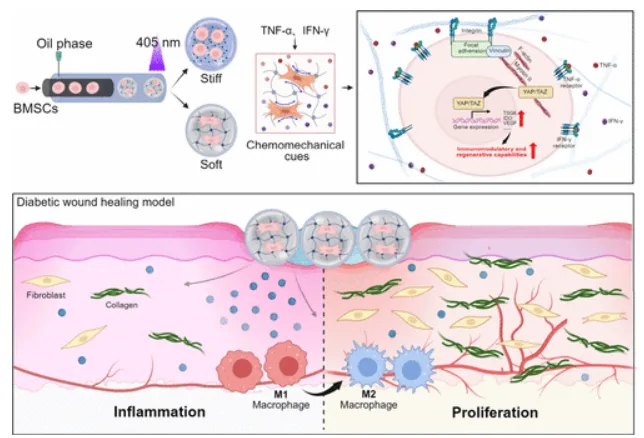
Overview of the study design. The top left shows MSC encapsulation in GelMA microspheres generated by a microfluidic process. The top right introduces the soft and stiff matrix conditions and inflammatory licensing with TNF-α and IFN-γ. The lower schematic links those chemomechanical cues to diabetic wound healing outcomes, including macrophage transition from M1 toward M2 and progression toward proliferation. Adapted from Hu W., et al. Synergistic Chemomechanical Cues within Mesenchymal Stromal Cell-Laden Hydrogel Microspheres for Accelerated Diabetic Wound Healing. 2026. ACS Applied Materials & Interfaces.
There is a useful parallel here with our earlier post on mesenchymal stromal cell spheroids, where the question was also less about whether MSCs are “good” in general and more about how their local context shifts the kind of repair signals they produce.
Why did hydrogel microsphere stiffness matter in this diabetic wound healing study?
The mechanical part of the study was built around two GelMA formulations with different degrees of methacrylation. Those differences produced two microsphere populations: a Soft group and a Stiff group. After fabrication, the authors ran uniaxial compression and reported average Young’s moduli of 2.45 ± 1.17 kPa for the Soft microspheres and 8.37 ± 2.95 kPa for the Stiff microspheres. They also used bulk hydrogel rheology as a supporting comparison, showing storage moduli around 250 Pa for Soft and 1,000 Pa for Stiff formulations.
That distinction is important because a lot of wound-healing biomaterials papers talk about “soft” or “stiff” constructs rather loosely. Here, the stiffness contrast was actually measured, and the paper relies on that measured difference all the way through the later biology. The microspheres were also highly monodisperse, with a selected diameter of about 229 ± 4 μm, which reduced some of the geometric variability that can otherwise muddy interpretation.
We have seen this same general idea in another application area before. In our post on tunable scaffold stiffness for in vitro liver models, stiffness was also treated as a biologically meaningful variable rather than just a material specification in the methods section.
How were MSC hydrogel microspheres for diabetic wound healing fabricated and measured?
The microspheres were fabricated by coaxial microfluidics. An aqueous GelMA precursor phase containing MSCs moved through the inner channel, while an oil phase flowed in the outer channel. Droplets formed at the junction and were crosslinked with 405 nm blue light to lock in the GelMA network. Microsphere size was tuned by changing the oil-to-aqueous flow ratio, then the authors chose a diameter that was practical for cell loading while still allowing diffusion through the construct.
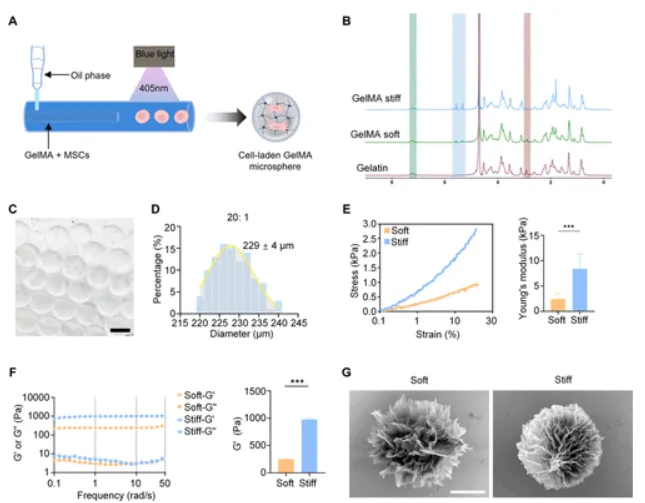
Fabrication and mechanical characterization of the GelMA microspheres. Panel A shows the coaxial microfluidic setup. Panels C-D show the monodisperse microsphere population selected for later cell work. Panel E is the key CellScale result: uniaxial compression measured on the MicroTester LT separated the Soft and Stiff groups by Young’s modulus. Panel G shows the porous microstructure of each formulation. Adapted from Hu W., et al. Synergistic Chemomechanical Cues within Mesenchymal Stromal Cell-Laden Hydrogel Microspheres for Accelerated Diabetic Wound Healing. 2026. ACS Applied Materials & Interfaces.
After fabrication, they did basic checks that matter for interpreting later biology. Brightfield images were used to confirm a tight size distribution. SEM showed a porous internal structure, with the softer formulation displaying larger pores. Even though stiffness is the headline variable, pore structure still affects transport and how cells engage with the matrix.
The authors then moved to compression testing, which is where the CellScale instrument enters the paper in a very direct way.
How did the MicroTester measure the mechanical difference between the microspheres?
This part is fairly straightforward, and that is useful. The compressive modulus of the GelMA microspheres was measured using a MicroTester LT, and the modulus was calculated from the initial linear region of the stress-strain curve from 0 to 20% strain.
That means the MicroTester was not a peripheral instrument in this workflow. It was the step that established the core experimental condition: whether the MSCs were sitting inside a softer or stiffer hydrogel microsphere. Without that measurement, the later claims about cell spreading, YAP localization, gene expression, macrophage polarization, and wound closure would be much harder to interpret.
In practical terms, the instrument was used here the way many groups use it in soft biomaterials work: to compress small, delicate samples directly rather than infer microsphere mechanics from bulk gel properties alone. The authors did include rheology on disc-shaped hydrogels, but the microsphere compression data are the more relevant measurement for what the cells actually experienced inside the carrier.
How did soft GelMA microspheres change MSC behaviour?
With the Soft and Stiff conditions established mechanically, the next step was simply to see what the MSCs did inside each niche. Viability stayed high in both groups (above 90% at days 1 and 3), so the comparison is not “good versus toxic.” It is a behaviour shift within a viable range.
By day 1, cells in the Soft microspheres looked less rounded and showed clearer spreading and protrusions. The quantitative shape metrics followed that: larger spreading area, higher aspect ratio, and lower circularity than the Stiff group. Vinculin intensity was higher in the Soft condition, and YAP showed greater nuclear localization as well. In other words, the softer microenvironment appeared to support stronger cell-matrix engagement and a more active mechanosensing state.
That is one of the moments where this paper shifts from a materials study to a mechanobiology study. The hydrogel is not just carrying the cells. It is changing how the cells interpret their surroundings.
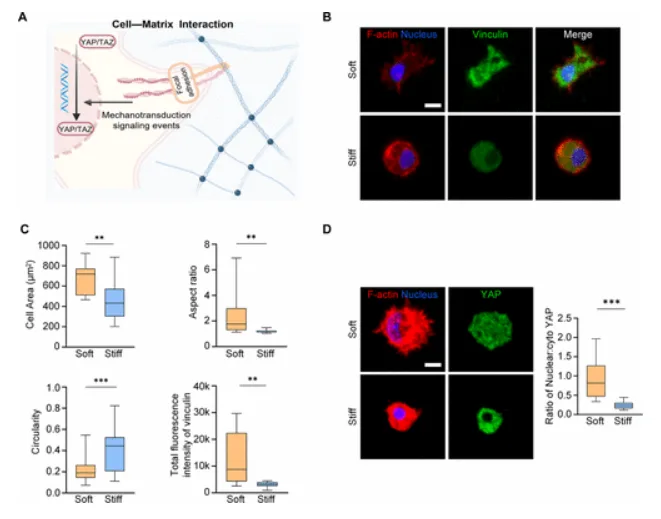
Early cell response inside soft versus stiff microspheres. Panel B shows representative fluorescence images of F-actin and vinculin, where the Soft group displays more cell spreading. Panel C quantifies spreading area, aspect ratio, circularity, and vinculin intensity. Panel D shows YAP localization, with higher nuclear translocation in the Soft group. Adapted from Hu W., et al. Synergistic Chemomechanical Cues within Mesenchymal Stromal Cell-Laden Hydrogel Microspheres for Accelerated Diabetic Wound Healing. 2026. ACS Applied Materials & Interfaces.
How did inflammatory priming and soft matrix mechanics work together?
The next question was whether that mechanically different state translated into a different therapeutic profile once MSCs were primed with inflammatory cytokines. The authors used TNF-α and IFN-γ as the priming signals and then measured gene expression associated with immunomodulation and wound repair.
Here the pattern gets more layered. Cytokine priming was still the main driver of the response overall, but the soft matrix amplified parts of that response. In the Soft group, the team saw increased expression of immunomodulatory genes including TSG6, IDO, COX2, IL-1ra, and CCL2, along with regenerative factors such as VEGF, TGFβ1, and FGF2. The authors describe the mechanical cue as a potentiator rather than a full replacement for biochemical licensing, which seems like the right way to frame it. The soft environment did not do everything alone. It changed how strongly the cells responded once the inflammatory signal was present.
They then pushed further into mechanism. Inhibiting YAP with verteporfin suppressed TSG6 expression, and inhibiting FAK with Y15 did the same. The data point toward a FAK-YAP mechanotransduction route being involved in how the soft microspheres shape the inflammatory response of the encapsulated MSCs.
Did the soft MSC hydrogel microspheres improve wound-related functions in vitro?
The authors did not stop at marker expression. They collected conditioned media from the microsphere cultures and asked what that media did to other cell types relevant to wound healing.
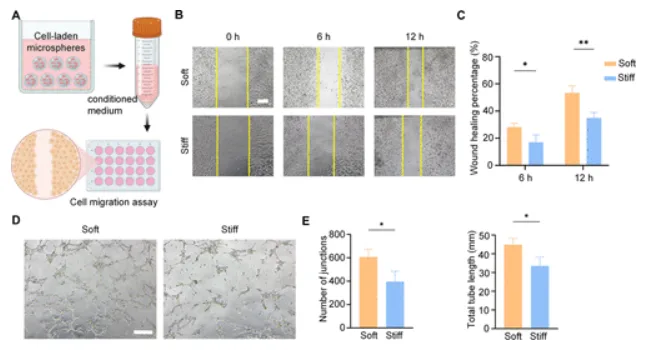
Functional readouts from conditioned media produced by the microsphere cultures. Panel B shows the scratch assay images at 0, 6, and 12 hours. Panel C quantifies faster closure in the Soft group. Panels D-E show the HUVEC tubule assay, where conditioned media from the Soft group produced more extensive vascular-like networks. Adapted from Hu W., et al. Synergistic Chemomechanical Cues within Mesenchymal Stromal Cell-Laden Hydrogel Microspheres for Accelerated Diabetic Wound Healing. 2026. ACS Applied Materials & Interfaces.
In a fibroblast scratch assay, conditioned media from the Soft group promoted faster migration. By 12 hours, scratch closure reached 53% in the Soft condition compared with 34% in the Stiff condition. In the endothelial assay, HUVECs formed more elaborate tubular networks when exposed to conditioned media from the Soft group, with greater branching and total tubular length.
They also tested immunomodulation more directly using a macrophage coculture setup. MSCs in soft microspheres reduced the proportion of pro-inflammatory M1 macrophages and increased the proportion of pro-regenerative M2 macrophages compared with the stiff condition. For diabetic wound healing, that shift may be one of the most consequential pieces of the paper, because chronic wounds are often defined by an inflammatory environment that fails to resolve on time.
For another wound-focused example of this broader theme, our post on oxygen-sensing 3D printed skin grafts also looks at how soft regenerative constructs are mechanically characterized before claims about wound function are made.
Did MSC hydrogel microspheres for diabetic wound healing improve repair in vivo?
The in vivo portion used a type 1 diabetic mouse wound model. After streptozotocin induction of diabetes, the researchers created standardized 8 mm full-thickness excisional wounds and compared three treatment groups: untreated control, acellular microspheres, and cell-laden microspheres containing cytokine-primed MSCs. Each wound received about 3,000 microspheres carrying a total of 6 × 10^4 MSCs.
The early wound images are probably the clearest place to start. By day 6, the cell-laden microsphere group had already moved past 50% wound closure, while the control and acellular groups were still around 20%. That gap is large enough that it is hard to dismiss as a small formulation tweak. Something about the cell-plus-matrix combination materially changed the healing trajectory in the early phase.
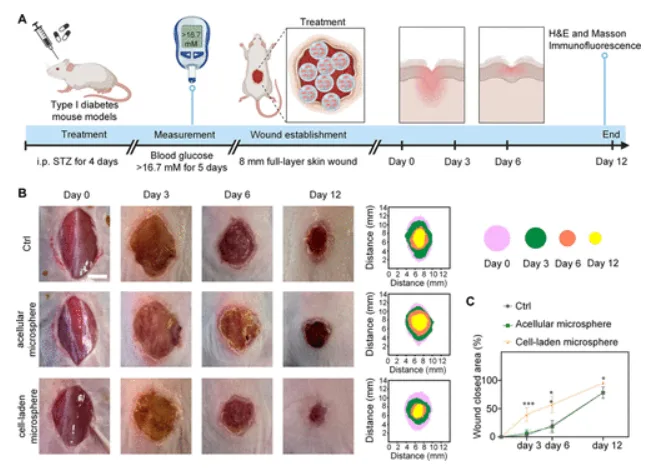
In vivo diabetic wound repair results. Panel A outlines the diabetic mouse wound model and treatment timeline. Panel B shows representative wound photographs and healing maps across days 0, 3, 6, and 12 for the control, acellular microsphere, and cell-laden microsphere groups. Panel C quantifies wound closure, with the cell-laden microsphere group separating early from the other treatments. Adapted from Hu W., et al. Synergistic Chemomechanical Cues within Mesenchymal Stromal Cell-Laden Hydrogel Microspheres for Accelerated Diabetic Wound Healing. 2026. ACS Applied Materials & Interfaces.
The histology supports that impression. Wounds treated with the cell-laden microspheres showed thicker neo-skin, more collagen deposition, and higher CD31-positive vessel density. Macrophage staining also shifted in the same direction seen in vitro: lower iNOS-associated M1 signal and higher CD206-associated M2 signal. The authors interpret that as immune reprogramming toward a more regenerative local environment, which seems reasonable given the rest of the data.
One thing I would keep a little guarded is that the in vivo comparison was focused on the selected best-performing formulation rather than a full soft-versus-stiff animal comparison. The paper itself notes that as a future direction. So the animal data are strong for the chosen treatment, but they do not fully isolate matrix stiffness in vivo the same way the in vitro experiments do.
What this study suggests about MSC hydrogel microspheres for diabetic wound healing
The broader point is not only that MSC hydrogel microspheres can support diabetic wound healing. It is that the local mechanical state of those microspheres may influence how the cells interpret inflammatory cues and what they release in response. In this paper, the softer GelMA condition was associated with more cell spreading, higher vinculin, more YAP nuclear localization, stronger upregulation of several therapeutic genes, improved fibroblast migration and endothelial tubule formation, a shift toward M2 macrophage polarization, and faster wound closure in diabetic mice.
That makes this a useful paper for anyone working on diabetic wound biomaterials, MSC delivery systems, hydrogel microspheres, or mechanobiology in regenerative medicine. It also fits into a larger pattern we keep seeing: if you do not measure the mechanics of the carrier, you end up guessing about one of the variables that may be driving the biology.
About the MicroTester
The CellScale MicroTester is a micro-mechanical testing instrument used to measure the force and deformation response of small, soft, and delicate samples. In this study, the MicroTester LT was used in uniaxial compression to quantify the stiffness of individual GelMA microspheres and calculate their compressive modulus from the initial linear region of the stress-strain curve. That measurement established the Soft and Stiff groups that the rest of the biological comparisons were built around.
For work like this, the value of the instrument is fairly practical. It gives researchers a way to test soft biomaterial constructs at the scale they are actually using, instead of relying only on bulk material assumptions. When the biological question depends on whether a microsphere is meaningfully softer or stiffer, that direct measurement becomes part of the study design rather than just a characterization add-on.