Drug-induced liver toxicity is one of the most common reasons promising compounds fail in development. That is one reason researchers keep looking for better in vitro liver models that can capture not just basic cell viability, but also disease-related changes in metabolism. In this Journal of Functional Biomaterials publication, researchers in Germany developed scaffolds with tunable stiffness to mimic both healthy and fibrotic liver tissue and then tested how those stiffness differences affected liver-cell behaviour.
Replicating liver stiffness is not a minor detail in tunable scaffold stiffness for in vitro liver models. Fibrosis and cirrhosis change the mechanical environment in ways that can shift hepatocyte function, including drug metabolism. The authors point out that if an in vitro model treats rigidity as irrelevant, it risks missing behaviour that matters in patients, especially when stiffness is part of the disease state.
Why scaffold stiffness matters in liver models
Hepatocytes and hepatocyte-like cells do not respond only to media composition. They also respond to the properties of the surrounding matrix. The paper reviews prior work showing that softer matrices can support a more differentiated and functional hepatic phenotype, while stiffer substrates can shift behaviour in less favourable directions. It also notes that liver stiffness below and above about 8 kPa is associated with different drug-metabolic enzyme activity in patients, which gives the scaffold-stiffness question real translational importance.
That is what makes this study more interesting than a simple scaffold-comparison project. It is really about how liver scaffold stiffness can be used to model disease-related differences in metabolism.
How the liver scaffolds were built
The researchers created pHEMA/BAA-based cryogel scaffolds and systematically varied composition to produce different physical properties. Four scaffold prototypes were compared, with attention to pore structure, porosity, permeability, water uptake, and stiffness. The production workflow is shown in the schematic in the publication, where the cryogel components are mixed, polymerized at low temperature, cut into slices, sterilized, pre-incubated, and then seeded with cells.
This part matters because the team was not just looking for any 3D scaffold. They were trying to find scaffold types that best matched the physical criteria needed for liver culture, especially pore architecture and disease-relevant stiffness.
What the mechanical testing showed
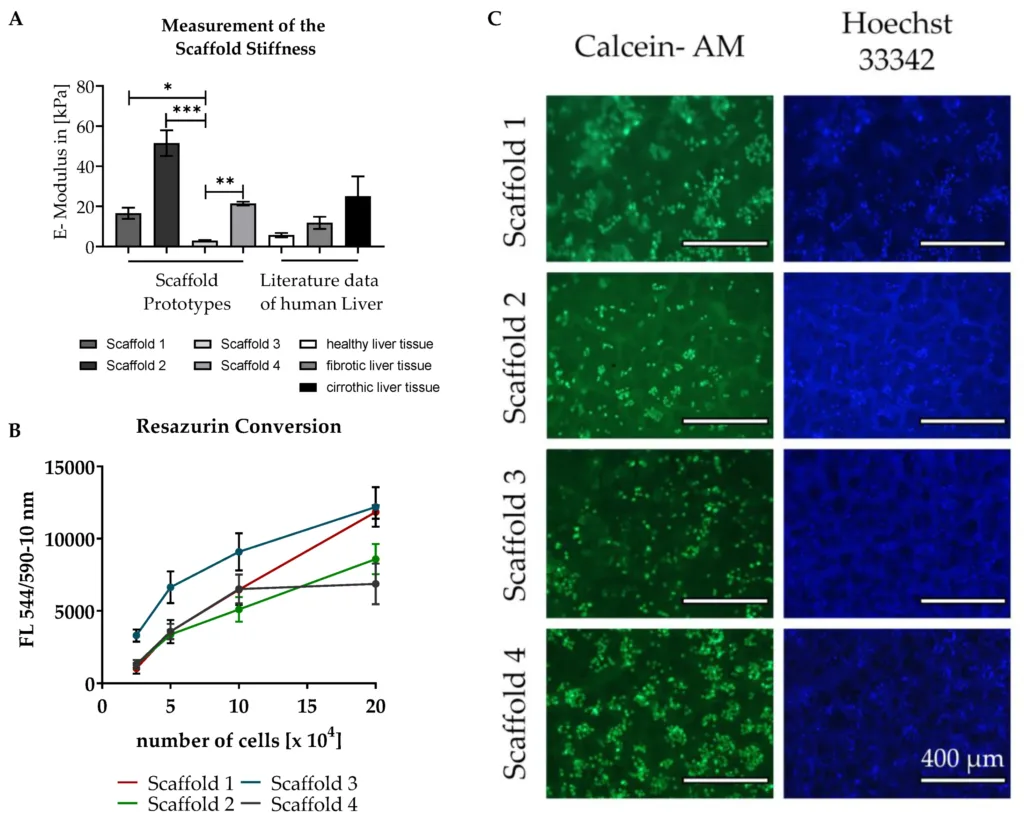
Adapted from Ruoß, M., Rebholz, S., Weimer, M., Grom-Baumgarten, C., Athanasopulu, K., Kemkemer, R., Käß, H., Ehnert, S., & Nussler, A. K. (2020). Development of Scaffolds with Adjusted Stiffness for Mimicking Disease-Related Alterations of Liver Rigidity. Journal of Functional Biomaterials, 11(1), 17. https://doi.org/10.3390/jfb11010017
To measure scaffold stiffness, the team used a CellScale MicroTester system (formerly MicroSquisher) in parallel-plate compression mode. Cylindrical samples about 3 mm high and 2 mm in diameter were compressed by 10% using a tungsten microbeam fitted with a 3 × 3 mm compression plate, and the resulting force-displacement data were used to calculate modulus.
The stiffness readout is what drives the rest of the paper. Prototype 3 came in at 2.9 ± 1.3 kPa, which the authors treated as a healthy-like target. Prototypes 1 and 4 landed in a stiffer range consistent with fibrotic or cirrhotic tissue. From there, the study essentially becomes a controlled comparison built around tunable scaffold stiffness for in vitro liver models: prototype 3 as the healthy-matched scaffold and prototype 1 as the main fibrotic-matched scaffold used in the downstream assays.
For readers who want more background on the method, see our page on Compression Testing. If your focus is broader material characterization, our Hydrogel Mechanical Testing content is also relevant.
Physical differences between the healthy and fibrotic-like scaffolds
Stiffness was the headline variable, but the scaffolds were not identical in everything else. The paper reports that prototype 1 had the largest pores (115 ± 29 µm), while prototype 4 was smaller on average (61 ± 41 µm). Prototype 1 also showed the highest water uptake and swelling, with prototype 3 next.
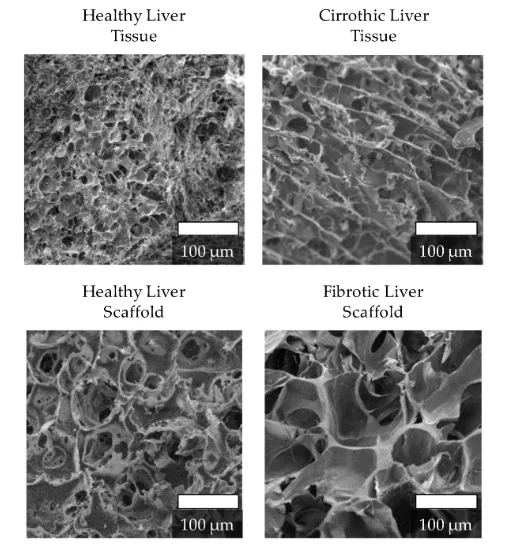
The imaging and porosity measurements add context for how “healthy-like” and “fibrotic-like” were implemented. The healthy-matched scaffold showed higher porosity, while permeability between the two selected scaffolds was reported as similar. The figure comparing native healthy and cirrhotic liver alongside the scaffold microstructures helps underline the point: the model is stiffness-guided, but transport and surface features are still part of what cells experience.
Pre-incubation had a major effect on cell attachment
One of the most useful practical findings in the paper was the effect of scaffold pre-incubation. Pre-incubating the scaffolds in FCS-containing medium significantly increased cell adherence, and the effect was especially pronounced in the healthy liver scaffold. The paper reports that pre-incubation increased cell attachment by as much as ten-fold in the healthy scaffold, compared with about 1.4-fold in the fibrotic scaffold.
The length of pre-incubation also mattered. Figure 6 shows that short pre-incubation periods of 24 to 72 hours led to relatively low attachment and viability after five days, whereas seven days or more produced much better outcomes. The Resazurin data and Calcein-AM images both support that pattern.
The authors also used SEM to examine the scaffold surfaces after pre-incubation. Figure 7 shows that the healthy liver scaffold developed large agglomerate-like deposits after seven days in FCS-containing medium, while the fibrotic-like scaffold surface changed much less. The paper suggests these surface changes may help explain the stronger cell-attachment effect.
What the metabolic results showed
Once the scaffolds were characterized, the authors tested whether stiffness affected liver-cell function. It did.
Cells plated on the softer healthy liver scaffold showed higher activity of CYP 3A4, CYP 2C9, and UGT compared with cells on the fibrotic scaffold. In contrast, CYP 1A2 and to some extent GST were higher on the fibrotic scaffold. There was no statistically significant difference in urea production between the two scaffold types.
That result is what makes the model especially interesting for drug studies. The system was not just able to distinguish soft and stiff scaffolds mechanically. It also produced stiffness-dependent differences in hepatic metabolism.
Why this matters for drug screening and fibrosis modelling
The value of the model is not simply that it created two scaffold types. The value is that it created a 3D liver culture system in which stiffness-dependent changes in metabolism can be studied under controlled conditions.
The scaffold-based 3D model mimics liver stiffness-dependent changes in drug metabolism and may help predict drug interaction in diseased livers more easily. That is a strong case for using tunable scaffold stiffness for in vitro liver models, especially in work related to fibrosis and toxicity screening.
For readers interested in broader model categories, see our pages on Organoid and Tissue Mimetic Systems and Drug Screening & Drug Delivery Mechanics.
Where the MicroTester fit into the study
The MicroTester was not just used for a one-off materials check. It played a central role in defining which scaffold formulations actually matched healthy versus fibrotic liver rigidity. Without that compression testing step, the biological comparison would have been much weaker.
Related reading
The full paper is available here: Development of Scaffolds with Adjusted Stiffness for Mimicking Disease-Related Alterations of Liver Rigidity.
If you want a related example of how small-scale mechanics are used in 3D culture systems, see our post on multiparametric analysis of tissue spheroids.
Final takeaway
This study is best understood as a scaffold-design paper with real functional consequences. The team did not just make liver scaffolds with different stiffness values. They showed that a softer scaffold matching healthy liver rigidity and a stiffer scaffold matching fibrotic liver rigidity could support measurably different metabolic behaviour, and that pre-incubation with FCS-containing medium strongly improved attachment, especially on the healthy-matched scaffold. That makes this a strong example of how tunable scaffold stiffness for in vitro liver models can improve disease-relevant drug metabolism research.