Tissue spheroids are becoming more common in preclinical research because they offer a more realistic model than simple 2D culture. They are used in drug screening, disease modelling, and cell-based assay development, but they also introduce a practical challenge. If spheroids are going to be useful as repeatable test systems, researchers need better ways to compare them from one sample to the next.
That is where micro-mechanical testing becomes valuable. Instead of looking only at morphology or molecular markers, researchers can also measure how a spheroid responds to compression. That adds a mechanical phenotype to the analysis, which can help distinguish cell populations, track maturation, and improve assay standardization.
Why stiffness matters in tissue spheroid analysis
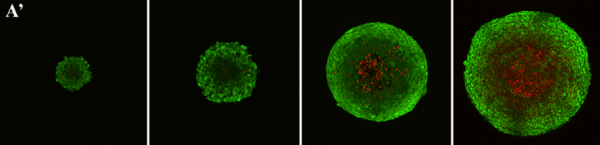
A spheroid may look visually consistent while still behaving differently under load. That matters because stiffness can reflect changes in cell packing, extracellular matrix development, phenotype, or overall aggregate organization. In other words, spheroid stiffness measurement can reveal information that may not be obvious from imaging alone.
This is one reason mechanical properties are a useful complement to molecular and structural markers. They do not replace those markers, but they add another layer of information. For researchers trying to compare spheroids across conditions, passages, or cell types, that extra layer can be very useful.
If you want a broader look at this application space, see our page on Microtissue & Spheroid Mechanics.
Micro-mechanical testing adds a direct physical readout
At the small scale, mechanical differences can be subtle. That is why low-force mechanical characterization is important. A suitable system needs enough sensitivity to test soft, small, and easily deformed samples without overwhelming them.
In this study published in the Biotechnology Journal, the researchers used mechanical properties as one of several metrics to characterize and evaluate tissue spheroids. The elastic moduli of cell aggregates were assessed using a CellScale MicroSquisher (now MicroTester), which supports the central point of the post: micro-scale stiffness measurement can serve as a useful analytical tool when comparing cell aggregates.
Why compression testing is a good fit for spheroids
Spheroids are soft, compact, and often close to spherical in shape, which makes micro-mechanical compression testing a natural method for analysis. A controlled compression test gives researchers a way to compare mechanical response across samples under defined conditions.
That matters for standardization. If two spheroids are intended to represent the same biological system but show very different compressive behaviour, that can indicate differences in internal structure or biological state. Used carefully, compression testing becomes part of quality control as well as characterization.
Why standardization matters in preclinical spheroid work
Spheroid-based assays are attractive partly because they can bridge the gap between simple culture systems and more complex in vivo models. But that value depends on consistency. A model that varies too much from one spheroid to another becomes harder to interpret in drug development or comparative biology studies.
Mechanical characterization helps with that problem because it gives researchers a direct, quantitative way to assess sample-to-sample variation. In that sense, micro-mechanical testing is not only a measurement technique. It is also a way to improve confidence in the assay itself.
For a related example of how mechanics can inform biologically relevant soft tissue models, read our article on Tumor Stiffness Is Driven by Cancer-Associated Fibroblasts and Collagen Remodeling.
How the MicroTester fits this type of work
The MicroTester is well suited for mechanical compression of tissue spheroids. These samples are small and mechanically delicate, so a system designed for micro-mechanical testing is a much better fit than a conventional bulk tester.
For readers working with small aggregates, spheroids, or similar engineered tissues, the value of the MicroTester is that it supports sensitive micro-scale stiffness measurement in a format matched to the specimen scale. That makes it useful for:
-
tissue spheroid mechanics
-
compression testing of small aggregates
-
low-force mechanical characterization
-
comparative cell-type analysis
-
assay standardization workflows
If your work involves similarly small or irregular specimens, you may also be interested in our article on Micro Testing Force in Irregularly Shaped Cell Clusters.
Publication Details
Read more about the tissue spheroid study here.
Final takeaway
Mechanical stiffness can add a useful phenotype, support better standardization, and help researchers compare spheroids in a more quantitative way. That makes it a strong fit for preclinical assay development and small-scale tissue characterization.