Tendon tissue engineering often looks straightforward on paper. Make an aligned scaffold, seed cells, add a differentiation cue, then wait for the construct to move in the right direction. In practice, it usually gets messier than that. Tendon cells and tendon-like cells do not just sit in a fibrous environment. They are pulled, relaxed, reoriented, and exposed to a relatively low-oxygen niche. Static culture can capture part of that picture, but not much of the mechanical part.
A recent study in Advanced NanoBiomed Research took that problem seriously. The authors built an aligned electrospun scaffold system with GDF-7-loaded mesoporous silica nanoparticles, then compared static culture with dynamically loaded culture under both atmospheric and physiological oxygen conditions. The dynamic loading step was done using the CellScale MechanoCulture T6, which the group used to apply controlled uniaxial strain to cell-seeded scaffolds over a 10-day culture period. What emerges from the paper is less a single dramatic result than a fairly convincing case that mechanical stimulation in tendon tissue engineering becomes more informative when it is combined with aligned topography, controlled oxygen tension, and sustained biochemical signalling.
The study focused on two cell populations: human mesenchymal stromal cells, or MSCs, and porcine tendon progenitor stem cells, or TPSCs. Both were seeded onto aligned polycaprolactone scaffolds that had been functionalized with GDF-7-loaded nanoparticles. From there, the authors looked at cell growth, scaffold mechanics, and tenogenic markers to see how far dynamic culture could push the system compared with static controls.
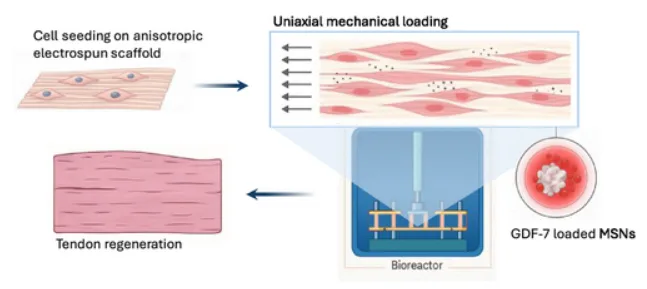
Overview of the tendon tissue engineering approach used in this study. The schematic shows the aligned electrospun scaffold functionalized with GDF-7-loaded mesoporous silica nanoparticles, followed by bioreactor-based mechanical stimulation to support cell alignment, matrix organisation, and tendon-like tissue formation. Adapted from VeraCitro, Aldo R.Boccaccini, Nicholas R.Forsyth, Tina P.Dale, Advanced NanoBiomed Research2026, 0, e202500218.
Why mechanical stimulation in tendon tissue engineering matters
There is already a good reason researchers keep returning to loading-based culture models in tendon work. Tendon is not simply a fibrous tissue with elongated cells. It is a mechanically active structure, and cell behaviour is tied closely to how strain is transferred through the matrix. The authors note that earlier tendon mechanobiology models have included 2D stretching systems, 3D scaffold systems, and ex vivo tendon loading models, but they also point out that these approaches do not always deliver multiple cues at once. That gap is what this paper tried to narrow.
That part is worth lingering on. One thing that stands out is that the paper is not really arguing for loading alone. It is arguing that mechanical stimulation in tendon tissue engineering may be more useful when it is placed in a setting that is already trying to look tendon-like in other ways. Here that meant aligned electrospun fibres, a sustained GDF-7 delivery strategy, and 2% oxygen as a more biorelevant culture condition. The mechanical input was one part of the environment, not the whole environment.
That broader context fits naturally with previous CellScale coverage of organ-on-a-chip mechanical stimulation methods and cyclic stretch and shear stress in vascular scaffolds, where the recurring theme is similar: once researchers begin trying to reproduce in vivo behaviour more faithfully, controlled mechanical cues stop feeling optional and start looking more central.
Building a more biorelevant tendon scaffold model
The materials side of the paper is fairly ambitious. The group synthesized mesoporous silica nanoparticles, loaded them with GDF-7, and incorporated those particles into aligned electrospun PCL fibres. Encapsulation efficiency was reported at 99.9%, and the release profile was designed to avoid a large burst release while extending delivery over several weeks. The scaffold itself was anisotropic, which matters in tendon studies because it gives the cells a directional cue before any external strain is applied.
There is a tradeoff here, though, and the paper does not hide it. Adding the nanoparticles reduced the baseline mechanical properties of the electrospun fibres. Young’s modulus and ultimate tensile strength both dropped after MSN incorporation, in both dry and wet conditions. That is probably not surprising if rigid particles disturb fibre structure or act as local stress concentrators, but it does matter because tendon scaffolds are usually judged partly by whether they can hold up mechanically while still functioning as a biologically active substrate.
This is part of what makes the later cell-driven mechanical recovery more interesting. The scaffold system did not start from a mechanically ideal state. It started from a state that had been altered by the delivery strategy.
How the MechanoCulture T6 was used in this study
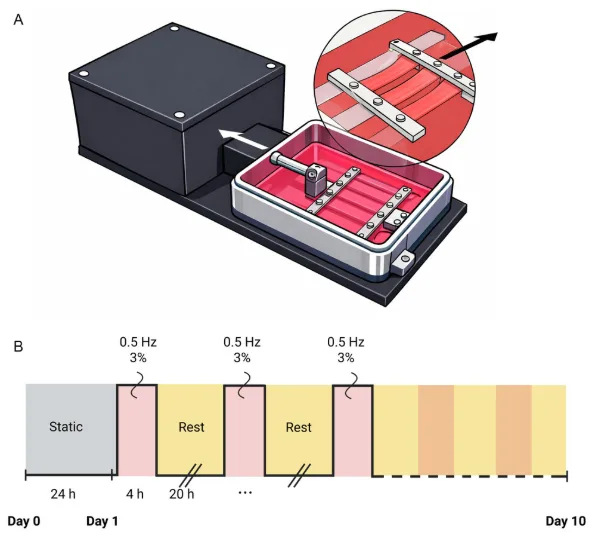
How the dynamic culture condition was created in this study. Panel A shows the MechanoCulture T6 setup used to hold and stretch parallel aligned scaffolds. Panel B shows the loading schedule applied after an initial static attachment period: 3% strain at 0.5 Hz for 4 hours per day over 10 days. Adapted from VeraCitro, Aldo R.Boccaccini, Nicholas R.Forsyth, Tina P.Dale, Advanced NanoBiomed Research2026, 0, e202500218.
The MechanoCulture T6 sits near the centre of the paper’s experimental design. After 24 hours of static culture to allow cell attachment, half of the seeded scaffolds were transferred into the bioreactor for dynamic culture. There, the constructs were stretched at 3% elongation, 0.5 Hz, 4 hours per day, for a total of 10 days. Static controls remained in standard well plates. The same study also compared culture at 21% oxygen and 2% oxygen, so the loading condition could be interpreted alongside oxygen tension rather than on its own.
That choice of loading regime appears deliberate. The authors were trying to stay within a range that was physiologically meaningful without pushing the scaffold-cell system into obvious damage or excessive detachment. In other words, the MechanoCulture T6 was not used here as a generic stretcher. It was used to create a repeated uniaxial loading environment that would let the researchers ask a fairly specific question: what happens when aligned tendon-like scaffolds are cultured under controlled cyclic strain rather than left static?
That also makes this study a nice companion to tendon scaffold mechanical testing under intermittent loading, because both pieces of work circle around a similar issue. Tendon scaffolds are rarely just passive materials. Their behaviour depends on what kind of loading history they experience.
Dynamic culture increased cell growth on aligned scaffolds
One of the clearer results in the paper comes from the cell-growth data. Live/Dead imaging showed viable cells across all groups, and the aligned scaffold topography already encouraged some directional organisation. Once cyclic loading was added, the alignment became more obvious, particularly for MSCs. The DNA quantification data then showed something a bit more concrete: dynamic culture produced roughly a two-fold increase in DNA content compared with static culture, and 2% oxygen generally pushed DNA content higher again.
That does not by itself prove tenogenic differentiation, of course, but it does suggest that the dynamic condition was not merely tolerated. The cells were responding to it, and in a way that seems consistent with a more supportive culture environment. For MSCs, the difference between static and dynamic culture became especially noticeable under 2% oxygen. TPSCs showed the same overall direction, although the oxygen effect was somewhat less uniform depending on condition.
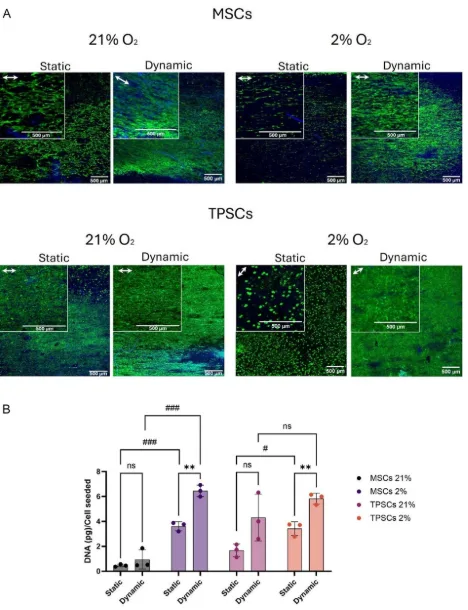
Dynamic culture increased cell growth on aligned scaffolds. Panel A shows representative Live/Dead images for MSCs and TPSCs under static and dynamic conditions at 21% and 2% oxygen. Panel B shows normalized DNA content, with higher values in the dynamically cultured groups and an additional benefit under 2% oxygen. Adapted from VeraCitro, Aldo R.Boccaccini, Nicholas R.Forsyth, Tina P.Dale, Advanced NanoBiomed Research2026, 0, e202500218.
Mechanical stimulation in tendon tissue engineering pushed cells toward a more tenogenic phenotype
The paper becomes more compelling when it moves from proliferation into phenotype. For MSCs, the early markers did not all move in a tidy upward direction, which is actually useful because it keeps the interpretation from becoming too neat. Mkx was downregulated in 3D culture relative to 2D controls, and Scx did not behave as a simple monotonic marker across all conditions. But Tnmd, a later tenogenic marker, told a clearer story. Under 2% oxygen, dynamic culture produced a marked increase in Tnmd expression relative to both 2D and 3D static groups.
TPSCs behaved somewhat differently, which makes sense given their more tendon-committed starting point. In 3D culture, they generally upregulated tendon-related genes, but the strongest Scx and Tnmd expression appeared when mechanical stimulation in tendon tissue engineering was combined with physiological oxygen. Under 2% oxygen, dynamic culture led to the highest observed levels of those markers, while Tnc-C followed a different pattern and did not rise in the same way.
That difference between markers is worth noting because it stops the paper from sounding like a universal “everything improved” result. It appears more accurate to say that cyclic loading plus physoxia particularly favoured the markers associated with a more mature tenogenic response, while some earlier or alternative markers were less consistent.
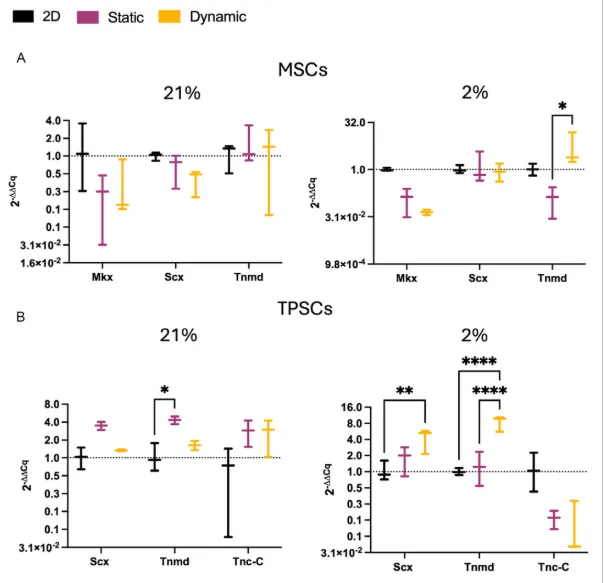
Mechanical stimulation and oxygen tension influenced tendon-related gene expression. Panel A shows Mkx, Scx, and Tnmd expression in MSCs under 2D, static 3D, and dynamic 3D culture at 21% and 2% oxygen. Panel B shows Scx, Tnmd, and Tnc-C expression in TPSCs under the same conditions, with the strongest Scx and Tnmd response appearing in the dynamically cultured 2% oxygen group. Adapted from VeraCitro, Aldo R.Boccaccini, Nicholas R.Forsyth, Tina P.Dale, Advanced NanoBiomed Research2026, 0, e202500218.
The immunofluorescence images make the response easier to see
The gene-expression data are important, but the immunofluorescence images help make the biological shift easier to interpret. In MSCs, cyclic loading increased Tnmd signal and produced visibly stronger actin alignment. TPSCs already showed higher baseline Tnmd expression, but even there the dynamic groups looked more elongated and aligned than the static ones. Under 2% oxygen, that combined effect appeared stronger again.
The cells in the loaded groups look more organised along the scaffold direction, and the actin orientation analysis showed a more pronounced preferred direction in dynamically cultured samples. That does not settle every mechanotransduction question, and the paper does not claim it does, but it does support the idea that mechanical stimulation in tendon tissue engineering is influencing structure as well as marker expression.
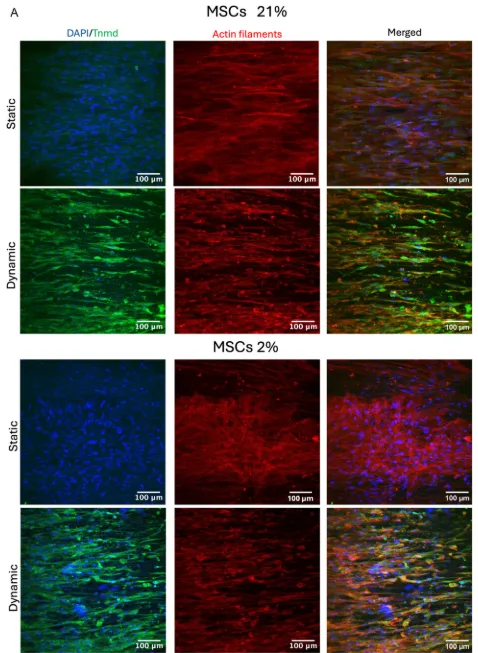
Dynamic loading changed how the cells looked as well as what they expressed. This image set shows MSCs under static and dynamic culture at 21% and 2% oxygen, with nuclei in blue, Tnmd in green, and actin filaments in red. The dynamically cultured groups show stronger alignment along the scaffold direction and a more visible Tnmd signal. Adapted from VeraCitro, Aldo R.Boccaccini, Nicholas R.Forsyth, Tina P.Dale, Advanced NanoBiomed Research2026, 0, e202500218.
Scaffold mechanics changed, then partially recovered after cell colonisation
A detail that could easily get lost in a faster summary is that the mechanical story has two phases. Before cell culture, the MSN-loaded scaffold was mechanically weaker than the particle-free version. After 10 days of culture, though, the cell-seeded constructs showed improved Young’s modulus and ultimate tensile strength compared with acellular controls. The effect was especially noticeable under 2% oxygen and in dynamically cultured groups, where the cells seem to have contributed enough matrix or structural reinforcement to offset at least part of the scaffold’s earlier weakness.
That makes the paper more interesting than a pure differentiation study. It suggests that scaffold conditioning and cell behaviour were not separate stories. They were coupled. The loaded scaffolds initially paid a mechanical cost for incorporating the growth-factor delivery system, then gained some of that performance back once the cells had time to colonise and reorganise the construct. In practical terms, this is the kind of result that makes bioreactor culture feel less cosmetic and more functionally relevant.
What this study says about the MechanoCulture T6
The MechanoCulture T6 was used here as a controlled uniaxial stimulation platform for flexible, cell-seeded scaffolds. In the context of this paper, that meant giving the authors a way to move beyond static incubation and ask how repeated strain changes cell growth, alignment, and tendon-related marker expression under defined culture conditions. The device was not the only important variable in the study, but it was the tool that created the dynamic environment the paper depends on.
More broadly, that is probably where the instrument fits best in tendon and scaffold work. It is useful when researchers want to impose a repeatable mechanical history on engineered tissues or soft biomaterial constructs rather than treating culture as mechanically neutral. This study used the system for tendon-like scaffolds, but the same general logic carries into other mechanobiology questions where static culture misses something important about how the tissue actually experiences its environment.
The paper stops where it should stop. It does not claim that tendon regeneration has been solved, and it does not jump from in vitro marker changes straight to clinical repair. What it does show is that when aligned scaffolds, sustained biochemical delivery, and low-oxygen culture are paired with controlled cyclic loading, the system moves in a more tendon-like direction. For researchers thinking about mechanical stimulation in tendon tissue engineering, that is a useful result, and for MechanoCulture users it is a good example of how the instrument is used in a study where loading is central to the biological question rather than added as an afterthought.