Resorbable vascular scaffolds are meant to disappear, but what happens while they are still there?. In vivo they sit in a mixed loading environment: the wall stretches with every pulse and flow adds shear at the surface. Cyclic stretch and shear stress can push biology in different directions, so the same scaffold can drive inflammation and remodeling, depending on how those cues play out.
In this Advanced Biosystems study, researchers from Eindhoven University of Technology examined how cyclic stretch and shear stress influence macrophage-fibroblast mediated tissue formation in an in vitro model of vascular regeneration. CellScale contributed to the study through the use of the BioTester, which was used for biaxial mechanical testing of the vascular scaffolds before and after culture. That mechanical testing helped connect changes in inflammation and matrix formation to functional changes in construct stiffness.
This paper is especially relevant for vascular tissue engineering because it shows that cyclic stretch and shear stress are not simply background conditions. They are active signals that influence mechanotransduction, immune response, and scaffold remodeling during tissue development.
Why cyclic stretch and shear stress matter in vascular regeneration
In this dataset, cyclic stretch and shear stress do not behave like interchangeable “dynamic culture” settings. Stretch is the one that drives the tissue-growth signal. Under cyclic stretch, the scaffolds show more proliferation and more matrix buildup than the static condition, and more than shear alone.
The matrix story follows the same pattern. With stretch present, the readouts shift toward a more matrix-producing state, including higher markers associated with collagen and elastic matrix development. The takeaway is simple: in this model, stretch is not just tolerated. It is one of the cues pushing the system toward neotissue formation early in culture.
For broader background on biomaterial mechanics in regenerative medicine, see our article on mechanical testing of biomaterials.
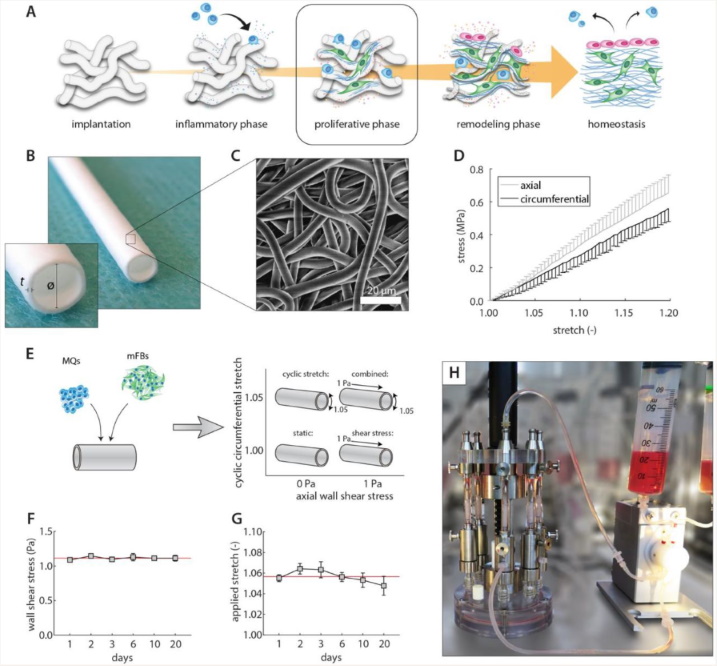
How the model was built
The researchers created electrospun vascular scaffolds from polycaprolactone bis-urea, or PCL-BU, a supramolecular elastomer relevant to cardiovascular biomaterials research. These scaffolds were seeded with a mixture of human primary monocytes and human vena saphena derived (myo)fibroblasts, then cultured for 20 days under controlled loading conditions.
The experimental design separated four conditions:
-
static culture
-
cyclic stretch alone
-
shear stress alone
-
cyclic stretch and shear stress together
This approach made it possible to compare how each loading mode influenced inflammation and tissue formation, both individually and in combination. That is important because vascular constructs are exposed to both forms of loading in vivo, not one in isolation.
Cyclic stretch promoted tissue growth
One of the clearest findings was that cyclic stretch and shear stress did not drive the same biological response. Cyclic stretch had the stronger effect on tissue growth. It increased cell proliferation and supported greater matrix accumulation within the scaffold, indicating that stretch acted as a powerful stimulus for new tissue formation.
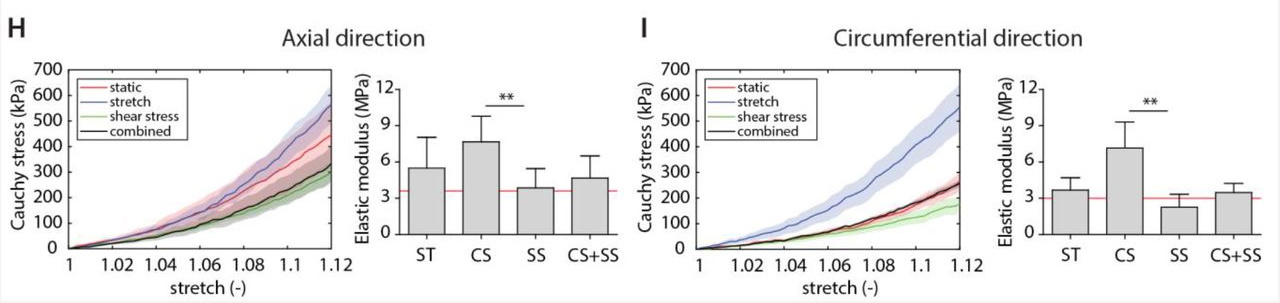
The matrix-related response was also notable. Stretch did more than increase cell number. Under cyclic stretch, the authors saw higher levels of markers tied to collagen and elastic matrix development, consistent with active matrix production inside the scaffold. For scaffold-guided regeneration, that is a useful reminder that cyclic deformation can act like a pro-growth cue early on, not just a stress the construct has to survive.
If you are interested in another example of mechanically active tissue models, our post on cardiac tissue disease modeling is also relevant.
Shear stress acted as a regulator of remodeling
While cyclic stretch promoted tissue growth, shear stress appeared to play a more moderating role. The study showed that shear stress influenced the inflammatory environment and helped regulate how much matrix accumulated under dynamic culture conditions.
That distinction matters. In a vascular graft context, more matrix is not automatically a win. If tissue builds too fast or remodels without control, the construct can stiffen in the wrong way or drift away from native-like mechanics. In this model, shear stress tempered some of the stretch-driven increases in matrix accumulation and stiffness, which fits with the idea that shear helps keep remodeling on a tighter leash.
This makes the paper especially useful for people interested in cyclic stretch and shear stress mechanobiology. It shows that different loading modes can push the same scaffold system in different biological directions.
For another cardiovascular mechanics example, see our blog on mechanical properties of heart valve leaflets.
How the CellScale BioTester was used
CellScale’s role in the study was to support the mechanical characterization of the scaffold constructs using the BioTester. The system was used for biaxial mechanical testing of vascular scaffolds, allowing the researchers to evaluate material stiffness in both axial and circumferential directions.
That detail is important because vascular tissues and vascular scaffolds do not behave the same way in every direction. Biaxial testing provides a more realistic view of mechanical behaviour than a single uniaxial measurement, especially for soft, anisotropic constructs that are meant to function under physiologically relevant loading.
In this study, the BioTester helped the researchers compare the mechanics of the original scaffold with the mechanics of the tissue-remodeled constructs after culture. That allowed them to directly link biological remodeling to functional mechanical outcome.

If you want a broader overview of this testing approach, our BioTester page and related application content on soft tissue mechanics provide more context.
Biaxial testing showed the mechanical consequence of remodeling
The mechanical results helped clarify the biological findings. Constructs exposed to cyclic stretch became stiffer, showing that the increased matrix formation had a measurable functional consequence. When shear stress was added alongside cyclic stretch, that stiffening effect was reduced.
That is one of the most useful takeaways from the paper. Shear stress did not simply add another source of loading. It changed the remodeling response in a way that limited the stiffness increase driven by cyclic stretch. For vascular scaffold design, that is a meaningful result because final construct mechanics are often just as important as early tissue formation.
This is exactly why biaxial mechanical testing of vascular scaffolds matters. It provides a way to evaluate how biological changes translate into directional mechanical behaviour, which is critical for cardiovascular biomaterials and engineered vascular tissues.
For a related post involving mechanically complex soft tissues, see venous valve tissue mechanical properties and ECM.
What this means for vascular tissue engineering
The main message of the study is that cyclic stretch and shear stress play distinct but complementary roles in scaffold-guided vascular regeneration. Cyclic stretch supported proliferation and matrix growth. Shear stress helped regulate that response and reduce excessive stiffening.
That balance is highly relevant to vascular tissue engineering, where success depends not only on producing tissue, but on producing tissue with the right composition, remodeling profile, and mechanical behaviour. A scaffold that fills quickly but remodels poorly may still fail. Studies like this one help explain why dynamic loading environments need to be designed with that balance in mind.
The work also reinforces a broader mechanobiology principle: loading conditions are not just test settings. They are part of the biological system.
Final thoughts
This study offers a strong example of how cyclic stretch and shear stress influence inflammation, matrix deposition, and construct mechanics in a vascular scaffold model. It shows that cyclic stretch can drive tissue formation, but that shear stress can help keep that response in check by promoting a more regulated remodeling environment.
For researchers developing regenerative vascular biomaterials, that is an important insight. The goal is not simply to maximize tissue growth. The goal is to guide tissue formation toward a stable, functional mechanical outcome.
You can read the publication here: Human In Vitro Model Mimicking Material-Driven Vascular Regeneration Reveals How Cyclic Stretch and Shear Stress Differentially Modulate Inflammation and Matrix Deposition
You can also read more about the research group here: Carlijn Bouten, Eindhoven University of Technology
For a different soft tissue loading mode, see our article on axial torsion of the annulus fibrosus.