For a while, cultured meat work was mostly framed around one question: how do you grow enough cells? That is still a major challenge, but it is no longer the only one. Once people start thinking about scale and actual food products, the material carrying those cells becomes part of the story too. It affects the culture process, the handling steps afterward, and in some cases the structure of the final product itself.
That is why cultured meat microcarriers have become such a useful area of focus. If the carrier can stay with the cells rather than being removed later, it starts doing more than one job. It helps with expansion, but it can also contribute to the final edible material.
Why cultured meat microcarriers matter
The value of cultured meat microcarriers is easiest to understand from a process point of view. Many of the cells used in cultivated meat need a surface to attach to while they grow. Standard microcarriers can provide that surface, but they often create extra work later because the cells still have to be taken off before the material moves into a food format.
Edible carriers change that logic a bit. If the carrier is already food-compatible, there is less pressure to separate it out at the end. The expanded cell material can move forward more directly, which makes the carrier useful not only during growth but also in product formation. That is why cultured meat microcarriers are starting to be treated less like temporary process aids and more like part of the scaffold strategy for the final material.
This is also where they begin to overlap with cultured meat scaffolds. The carrier is not only there to support cell expansion anymore. It can influence texture, cohesion, and appearance once the cells are turned into something more structured.
Edible microcarriers in cultured meat production

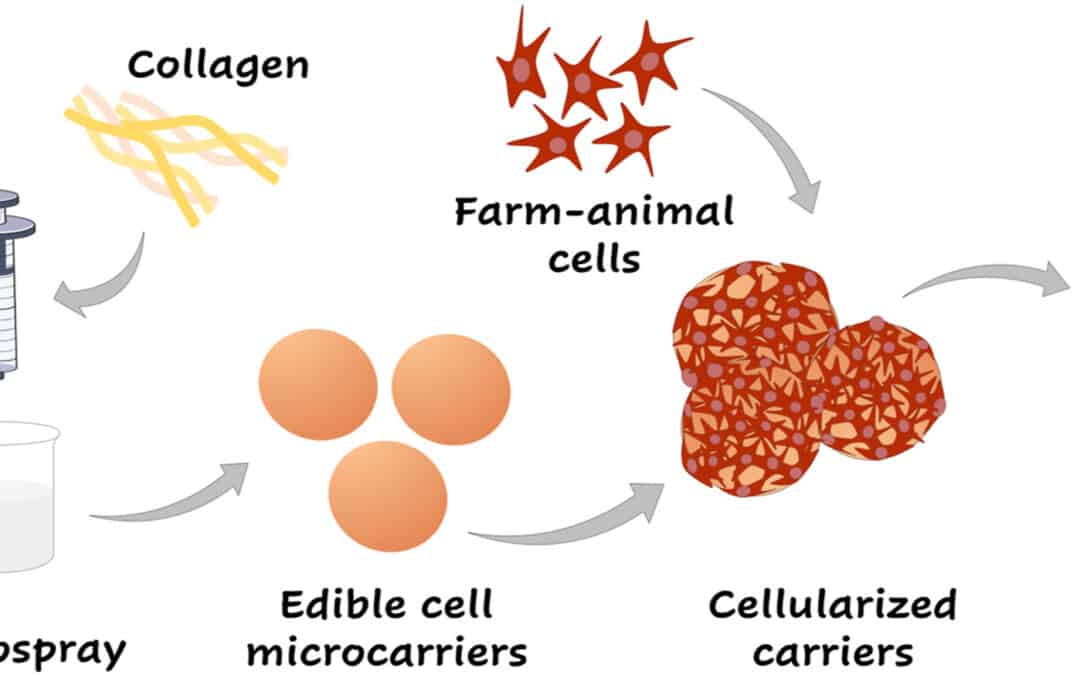
A strong example comes from work on edible microcarriers made from shellfish-derived chitosan and collagen. In that cultured meat study, bovine mesenchymal stem cells were expanded on edible carriers in a scalable bioreactor workflow, then used as cellularized microtissues that could be assembled into different cultured meat formats. The microcarriers were not treated as a disposable support. They were designed to stay in the product.
That is what makes edible microcarriers so useful in cultured meat production. They can:
-
support cell attachment and expansion
-
reduce the need for harvesting steps
-
remain in the final product
-
contribute to texture and structure
-
add nutritional value depending on composition
In this case, shellfish-derived chitosan and collagen were not only food-compatible. They also created a mechanically relevant support for large-scale cell culture and later aggregation.
Mechanical testing of cultured meat microcarriers
Mechanical characterization of cultured meat microcarriers is essential for ensuring structural integrity during large-scale cell expansion, processing, and final product formation in cultured meat systems.
If the carriers are too weak, they may not hold up during culture and handling. If they are too soft or too brittle, they may not contribute anything useful to the final texture. If their mechanics change too much during cell growth, the process becomes harder to control.
In the cultured meat platform study mentioned above, the researchers used a CellScale MicroTester G2 to measure the mechanics of the microcarriers, the cellularized microtissues, and the larger aggregated structures they formed later. That kind of micro-mechanical testing is especially relevant here because the materials are small, soft, and structurally delicate.
Micro-mechanical testing and stress-strain analysis
For cultured meat microcarriers, bulk mechanical data are not always enough. The structures are small, highly hydrated, and often change significantly as cells grow and produce their own matrix. That is why micro-mechanical testing and stress-strain analysis are so useful.
These methods can help show:
-
how much force a microcarrier can tolerate
-
how it deforms under load
-
whether the material remains stable during cell expansion
-
how much stiffer the system becomes after cells form aggregated microtissues
-
whether a carrier is likely to support later product structuring
In the chitosan-collagen study, that mechanical shift was substantial. The resulting disc-like aggregates reached a Young’s modulus of about 80 kPa, roughly 40 times higher than the microcarriers or microtissues alone. That result shows why stress-strain analysis matters. The system does not stay mechanically static as the biology develops.
Cultured meat scaffolds and final product structure

The conversation about cultured meat microcarriers naturally leads into cultured meat scaffolds, because once the cells have expanded, the question becomes how to organize them into something food-like.
A slurry of cells is not enough. To build a structured product, the material needs some combination of cohesion, stiffness, softness, and shape retention. That can come from aggregated microtissues, larger edible scaffolds, or both.
The cultured meat paper above makes this practical by showing different final formats built from cellularized microtissues, including a layered product and a burger-like product with oleogel-based fat substitution. Those final structures did not have the same mechanical feel. The layered version was stiffer, while the burger-like version was softer and more marbled in appearance.
That is exactly why cultured meat scaffolds and cultured meat microcarriers should be discussed together. The upstream support material affects the downstream food structure.
Compression testing of edible microcarriers and microtissues
When working with soft food-grade materials, compression testing often gives more useful application data than abstract material description alone. For edible microcarriers and cellularized aggregates, compression-style measurements can help estimate whether the material will hold shape, deform too easily, or begin to approach a more realistic food texture.
That is particularly relevant in cultured meat, where the material is not being designed only for cell compatibility. It is also being developed for handling, assembly, and eating quality.
For these systems, compression testing can help answer:
-
does the material collapse too easily
-
does cell growth stiffen the structure in a useful way
-
can the microtissues be combined into larger formats without losing integrity
-
does the scaffold contribute to bite or cohesion in the final product
Hydrated biomaterial testing in cultured meat systems
Most cultured meat microcarriers and cultured meat scaffolds are soft and water-rich. Their behaviour changes a lot depending on testing conditions. That is why hydrated biomaterial testing should be part of the conversation.
Testing a soft edible scaffold in a dry or unrealistic state can make the numbers look cleaner than they really are. But the process itself happens in aqueous, cell-culture conditions, and the final mechanics during growth are influenced by hydration, matrix swelling, and biological remodeling.
That is one reason hydrated biomaterial testing and low-force mechanical testing matter so much here. They produce data that are more relevant to how the material actually behaves during expansion and product formation.
Cultured seafood scaffolds show the same pattern
This is not only true for mammalian cultured meat. The same pattern appears in cultivated seafood.
In a fish study on edible electrospun fibers designed to mimic myoseptum-like structure, the researchers mechanically tested the scaffold materials using a CellScale UniVert. They found that thermal crosslinking increased elastic modulus 1.90-fold and ultimate tensile strength 1.8-fold. The fibers also supported fish-cell attachment and proliferation, while aligned fibers improved cell alignment.
Whether the material is an edible microcarrier or an edible fibrous scaffold, the same principle holds: if the mechanics are wrong, the biological and structural outcome will suffer.
Why cultured meat microcarriers matter for scale-up

One of the hardest parts of cultured meat commercialization is that the system has to work as biology, process engineering, and food structure at the same time. Cultured meat microcarriers are valuable because they touch all three.
They support large-scale expansion.
They reduce downstream processing.
They can stay with the cells and become part of the final product.
They influence texture and structure later on.
But all of that only works if the carrier is mechanically fit for the process. A carrier that is food-safe but mechanically weak is still a poor carrier. A carrier that supports cell growth but creates the wrong final texture is also limited. That is why mechanical testing belongs in the development workflow from the beginning.
Conclusion
Cultured meat microcarriers are helping cultivated protein systems move beyond simple cell expansion by acting as both growth supports and edible building blocks. In food-grade systems based on materials such as chitosan and collagen, these edible microcarriers can reduce harvesting steps, support scale-up, and contribute directly to final product structure.
At the same time, the literature makes it clear that micro-mechanical testing, compression testing, stress-strain analysis, and hydrated biomaterial testing are all important for understanding whether those materials are physically suitable for real cultured meat workflows. Mechanics affect cell expansion, aggregation, processing, and texture. That is true for microcarriers, and it is true for larger cultured meat scaffolds as well.
Related Reading: The CellScale MicroTester has the perfect 50 micron to 5 millimeter scale needed for researchers at Georgia Tech. Their samples are not tissues, or single cells but rather micromass aggregates of stem cells. Learn how our technology surpasses Instron machines or atomic force microscopy to get the mechanical properties of their samples.