Scanningless 3D Bioprinting of Human Tissues with dECM Bioinks
Decellularized extracellular matrices (dECM) are widely used in regenerative medicine, cancer research, drug development (and many more areas) primarily because they preserve many of the biochemical and structural cues found in native tissues. When you remove cellular components while retaining extracellular matrix architecture, dECM provides a biologically relevant scaffold for engineering functional human tissues.
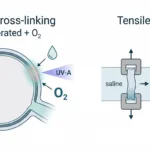
In a peer-reviewed study published in Biomaterials, Dr. Claire Yu and colleagues at the University of California, San Diego reported a scanningless and continuous 3D bioprinting approach for fabricating human tissues (using photocrosslinkable, tissue-specific dECM bioinks). Their work (using the MicroTester) demonstrates how rapid bioprinting combined with microscale mechanical testing enables precise control of tissue stiffness in engineered cardiac and liver models.
Why Mechanical Stiffness Matters in 3D Bioprinted Tissues
In engineered tissues, it isn’t just biochemical composition that determines function. Mechanical properties (like stiffness for example) play a critical role in regulating cell behaviour, maturation, and long-term viability. Matching the mechanical environment of native tissue is especially important for applications such as cardiac tissue engineering, liver disease modeling, and cancer research.
This study highlights why directly measuring stiffness is critical in 3D biological models, particularly when small changes in matrix composition can produce large functional differences in tissue mechanics.
Why directly measuring stiffness is critical in 3D biological models
Scanningless Bioprinting with Tissue-Specific dECM Bioinks
The authors developed photocrosslinkable dECM bioinks derived from tissue-specific extracellular matrix. Using a digital light processing (DLP)-based 3D bioprinter, they fabricated complex, high-resolution tissue structures without the need for scanning or layer-by-layer rastering.
This scanningless approach enabled:
-
Rapid fabrication of tissue constructs
-
Precise microscale patterning
-
High cell viability during and after printing
The resulting cardiac and liver tissue models exhibited organized cellular architecture and supported tissue-specific cell maturation.
Mechanical Tuning Validated with Microscale Compression Testing
To quantify mechanical performance, the research team used the CellScale MicroTester to measure the compressive Young’s modulus of the printed dECM hydrogels. By applying controlled micro-compression tests, they directly linked printing parameters (such as exposure time and bioink formulation) to measurable changes in stiffness.
This approach allowed the researchers to:
-
Tune stiffness across physiologically relevant ranges
-
Match mechanical properties of native liver and developing heart tissue
-
Decouple visual appearance from mechanical function
The study demonstrates how connecting matrix composition to measurable mechanical function is essential for validating engineered tissue models and interpreting biological outcomes.
Explore the characterization of crystalline austenitic stainless steel using the CellScale UniVert here.
Linking Bioprinting, Mechanics, and Disease Modeling
By integrating rapid 3D bioprinting with direct mechanical testing, this work establishes a framework for building tissue models where stiffness is not inferred, but measured. This is particularly relevant for disease modeling applications (i.e., fibrosis, cancer mechanobiology, drug response studies) where extracellular matrix mechanics strongly influence cellular behavior.
The combination of dECM bioinks and microscale mechanical characterization provides researchers with a powerful toolset for engineering tissues that are not only biologically relevant, but mechanically accurate.
Read the Full Publication
Yu, C. et al. Scanningless and Continuous 3D Bioprinting of Human Tissues with Decellularized Extracellular Matrix. Biomaterials, 2019. https://doi.org/10.1016/j.biomaterials.2018.12.009.
Explore more CellScale-enabled research at: https://www.cellscale.com/publications/