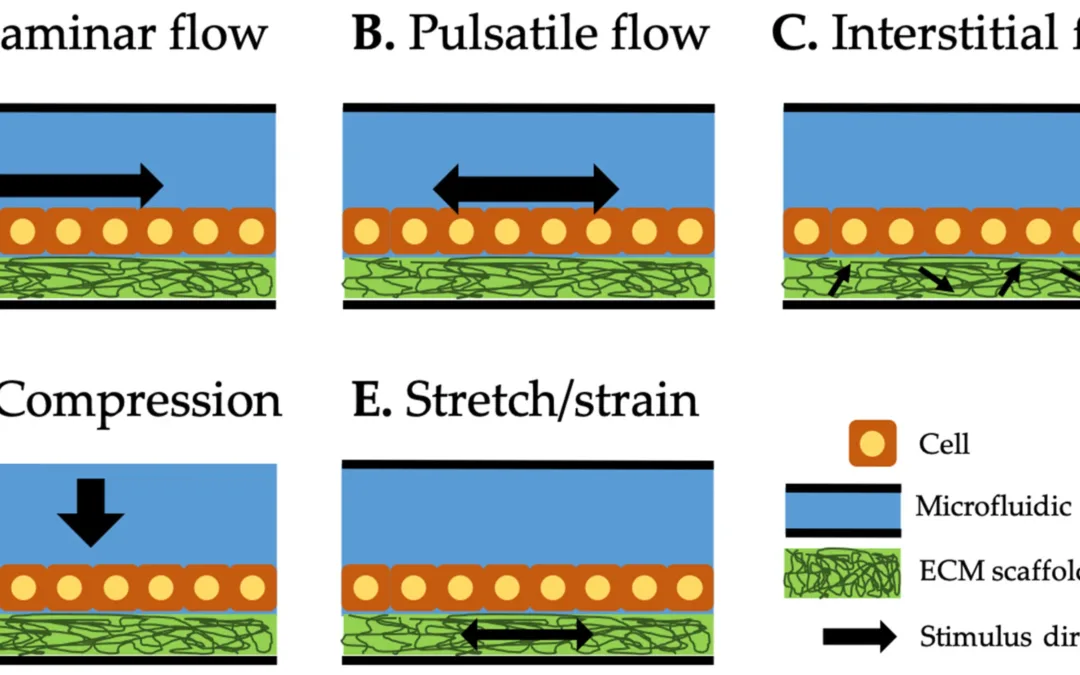
Organ-on-a-chip mechanical stimulation has become one of the most important design considerations in microphysiological systems. Building a chip with the right cells is only the starting point. If the goal is tissue-like behaviour, the culture has to be loaded in tissue-like ways. In practice that means adding forces such as flow shear, compression, and cyclic stretch or strain, depending on the organ model. Those inputs can change how cells organize, migrate, differentiate, and signal through mechanotransduction pathways.
That is the central message of this review paper from Kattika Kaarj and Jeong-Yeol Yoon at the University of Arizona. The paper surveys the main ways researchers deliver mechanical forces in microphysiological systems and explains why these inputs matter for organ-level function, disease modelling, drug development, and toxicology.
CellScale is mentioned directly in the stretch and strain section, where the review highlights the MechanoCulture B1, BioTester, and MechanoCulture T6 as examples of systems used in mechanobiological studies.
For a broader introduction to the field, see our educational post on heart-on-a-chip.
Why organ-on-a-chip mechanical stimulation matters
The review keeps returning to a basic point: cells do not develop in a mechanically quiet environment. In vivo they are always being pushed, stretched, or sheared, and they use those cues to regulate function. Organ-on-a-chip mechanical stimulation is one of the ways microphysiological models move closer to that reality. The authors tie this directly to mechanotransduction, since the whole premise is that mechanical signals are converted into biological responses that shape phenotype.
They also make it clear this is not limited to one niche application. The same logic shows up across wound healing, fibrosis, cancer, vascular models, and lung biology. In other words, organ-on-a-chip mechanical stimulation is not an optional add-on. It is often the difference between a static culture device and a model that behaves more like tissue.
The three main categories of mechanical stimuli in OOC systems
One of the strengths of the review is that it organizes mechanical stimuli in organ-on-a-chip systems into three main categories: shear flow, compression, and stretch/strain. Shear can be delivered as laminar, pulsatile, or interstitial flow. Compression applies force normal to the cell layer or construct. Stretch and strain apply tensile deformation to the substrate, scaffold, or tissue analogue.
That framework is useful because it gives readers a practical way to think about stretch and shear in OOC devices. Different tissues experience different combinations of these forces, and the challenge in OOC design is often deciding which ones matter most for the biology being modelled.
The review also makes clear that some of the most physiologically relevant systems combine more than one input. Lung-on-a-chip and glomerulus-on-a-chip examples in the paper integrate both flow and cyclic deformation, which better reflects how those tissues function in vivo.
Shear flow in organ-on-a-chip systems
Shear flow is described in the review as the most common mechanical stimulus used in OOC systems. It arises when liquid moves through microchannels and applies tangential stress to cells cultured on channel surfaces or within extracellular matrix environments. For shear, the review separates three common modes: laminar flow, pulsatile flow, and interstitial flow. They matter for different reasons depending on the tissue you are trying to mimic.
Vascular and kidney examples are the obvious cases, since flow changes morphology, transport behaviour, and endothelial function. The paper also points out that interstitial flow shows up in cancer and angiogenesis models, where it can shift invasion behaviour and sprouting. The thread that ties these together is that flow is doing more than moving media. It is one of the core mechanical inputs in many microphysiological systems.
Compression in organ-on-a-chip systems
Compression is another important category, particularly for tissues such as bone and cardiac constructs. The review describes systems where compressive loads are applied through pistons or pressure-controlled diaphragms, allowing researchers to study cell responses to mechanical loading in a more tissue-relevant way. These models have been used to investigate osteogenic differentiation, intracellular mechanostress responses, and cardiac tissue formation under compression.
This section is useful because it broadens the conversation beyond flow. When people think of OOC devices, they often picture perfused microchannels first. But the review shows that organ-on-a-chip mechanical stimulation also includes direct compressive actuation when the target biology calls for it.
Stretch and strain in OOC devices
The stretch and strain section is where CellScale becomes even more relevant. The review explains that cyclic strain can be generated in OOC models through flexible substrates, vacuum-driven membrane deflection, or other forms of actuation that deform the culture environment. Lung-on-a-chip systems are a prominent example, where cyclic strain is used to mimic breathing-related motion.
The broader message is that organ-on-a-chip stretch and strain can be just as important as flow. In tissues that naturally expand, contract, or deform cyclically, static culture misses a major part of the biological environment. That is why stretch-based OOC models are so important for mechanobiology and translational in vitro research.
For a related example of disease modelling in dynamic tissue systems, see our post on cardiac tissue disease modeling.
How CellScale systems were used
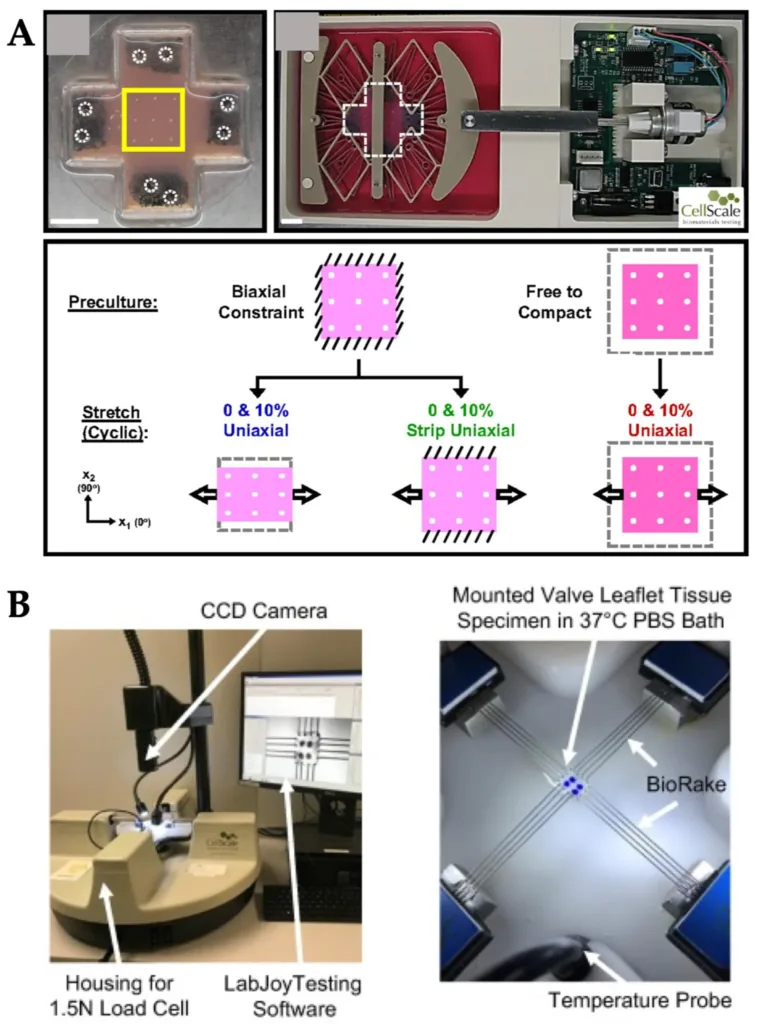
CellScale systems used for stretch and strain studies in organ-on-a-chip and mechanobiology research. Adapted from Kaarj, K.; Yoon, J.-Y. Methods of Delivering Mechanical Stimuli to Organ-on-a-Chip. Micromachines 2019.
CellScale is specifically mentioned in the review as part of the growing set of commercial tools used to deliver stretch and strain in mechanobiological studies. The paper highlights three separate examples:
The MechanoCulture B1 was used for cyclic uniaxial stretch experiments in 2D and 3D fibroblast culture, where cyclic stretch induced perpendicular fibroblast alignment in 3D culture.
The BioTester was used to apply biaxial stretch to porcine atrioventricular heart valve leaflets so researchers could characterize regional heterogeneity and function under different loading ratios and stress-relaxation protocols.
The MechanoCulture T6 was used to apply cyclic radial strain to mitral valve anterior leaflets to study how the tissue responded to physiological stretch.
This is a valuable point for the blog because it shows that CellScale’s role in this area spans both mechanical stimulation and mechanical testing. In OOC and mechanobiology work, stimulation is used to create a physiological loading environment, while testing is used to characterize how tissues or constructs respond. The review positions CellScale systems within both kinds of workflows.
For more on one of the cited tissue applications, see our post on mechanical properties of heart valve leaflets.
Mechanical stimulation versus mechanical testing
This distinction is worth making clearly for readers. Organ-on-a-chip mechanical stimulation is about recreating the forces cells experience in vivo so that culture conditions better reflect physiology. Mechanical testing, by contrast, is about measuring tissue or material response under defined loading. In practice, modern research often brings these together. A platform may stimulate a construct during culture, then test tissue response, remodelling, or directional mechanics afterward.
Multiple forces often need to be combined
A particularly strong point in the review is that single-force models are often not enough. Some tissues naturally experience multiple mechanical inputs at once. The lung is one example, where airflow, vascular flow, and cyclic deformation all matter. The paper also discusses vascular and angiogenesis models where both luminal shear and interstitial flow contribute to the biological response.
That makes organ-on-a-chip mechanical stimulation a design problem as much as a biological one. Researchers are not only asking which cells to include. They are also deciding which combinations of forces must be recreated to produce meaningful tissue-level behaviour.
Why this review matters for mechanotransduction research
The review is especially strong for mechanotransduction audiences because it keeps returning to the idea that mechanical cues are not secondary variables. They are central regulators of cell and tissue behaviour. The paper links mechanical stimulation to endothelial sprouting, epithelial barrier function, fibroblast alignment, cardiac tissue engineering, and mechanobiological imaging workflows.
It also points toward the next step for the field: more complex systems that combine multiple stimuli and support real-time monitoring. The review discusses imaging-enabled OOC platforms and notes that real-time observation can improve mechanobiological studies by showing how cells respond dynamically to changing load.
Final thoughts
This review makes a strong case for why organ-on-a-chip mechanical stimulation deserves more attention in both OOC design and biomaterials research. Shear, compression, and stretch are not interchangeable inputs. They recreate different aspects of the in vivo environment and can produce very different biological responses. For researchers building more physiologically relevant microphysiological systems, understanding those differences is essential.
It is also encouraging to see CellScale systems featured directly in this context. The review cites the BioTester benchtop biaxial tester and our MechanoCulture bioreactors as tools used in mechanobiological studies involving fibroblast alignment, heart valve leaflet mechanics, and mitral valve stretch response. That places CellScale within the broader conversation around mechanical forces in microphysiological systems and organ-on-a-chip mechanotransduction.
Read the full review here: Methods of Delivering Mechanical Stimuli to Organ-on-a-Chip
For related reading, you may also like: