Heavy metals such as copper are persistent pollutants in industrial wastewater, which is why there is so much interest in adsorbent materials that are both efficient and recyclable. Researchers at the University of Waterloo developed cellulose aerogel beads for copper removal that were not only effective adsorbents, but also mechanically robust under wet testing conditions. That second point matters more than it may seem at first. If an adsorbent falls apart in water, its chemistry may be promising, but its practical usefulness drops quickly.
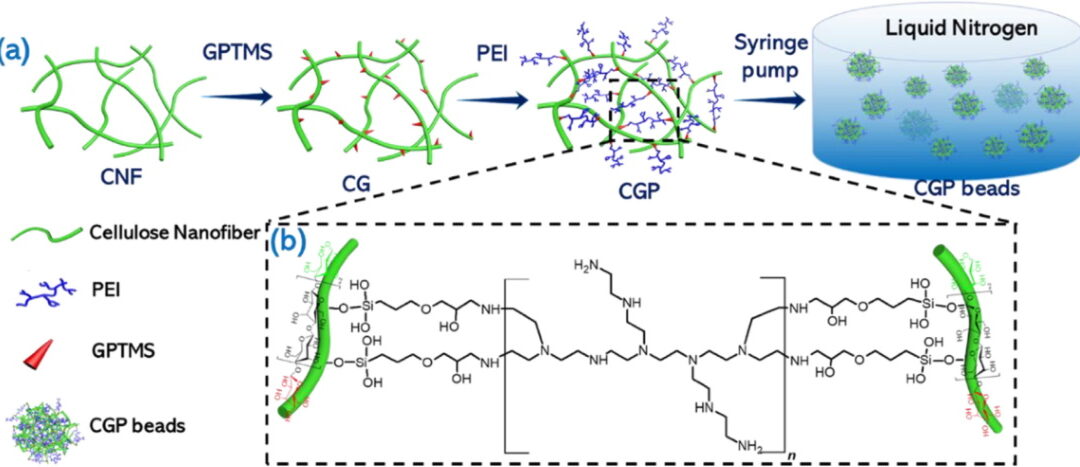
The team used cellulose nanofibrils as the scaffold, then cross-linked polyethylenimine onto the network using GPTMS to create small porous beads with high amine content. The paper shows that these cross-linked beads were designed to solve several problems at once: improve copper adsorption, shorten diffusion path length, increase wet mechanical strength, and support regeneration and reuse.
Why cellulose aerogel beads are useful for copper removal
Many adsorbents for heavy metal removal are powders. The paper points out the usual drawbacks: poor recyclability, lower removal efficiency, and the risk of secondary pollution. Aerogels are attractive because they combine high porosity, high surface area, and easier separation from bulk aqueous solution. In this case, using cellulose aerogel beads instead of larger monoliths also helped improve adsorption speed by reducing diffusion path length.
That design choice is one of the best parts of the study. The authors were not only looking for high adsorption capacity. They were also trying to build a format that would work better in practice, especially for applications where recyclability and flow-through handling matter.
How the beads were made
The researchers cross-linked branched PEI onto cellulose nanofibrils using GPTMS, then injected the mixture into liquid nitrogen to form spherical frozen droplets before freeze drying. The result was a bead format roughly 3 to 4 mm in diameter, with a porous internal structure and high amine content for copper binding. The schematic on page 4 shows the process clearly, from CNF and GPTMS through PEI addition and bead formation in liquid nitrogen.
The chemistry mattered because the amine-rich network was central to the adsorption mechanism. Increasing the PEI:GPTMS ratio increased total amine content and improved copper uptake. The paper reports that total amine content rose from 2.67 mmol/g in CGP1 to 5.74 mmol/g in CGP3, which is part of why the higher-PEI beads performed better.
Why mechanical testing mattered in this study
The authors did not treat mechanical testing as a side note. They used compressive stress-strain measurements to evaluate whether the aerogel beads remained structurally stable in water and during adsorption. Individual beads were tested with a MicroTester G2 using a 5 mm × 5 mm steel platen in a water bath, with displacement and force recorded in real time.
That is important because unmodified CNF aerogel beads readily disintegrated in water, while the cross-linked beads remained intact. The paper directly links that improvement to robust covalent cross-linking, showing that chemistry and mechanical integrity were tightly connected in the final material design.
For readers who want more background on the method, see our page on Compression Testing.
What the compression results showed
The compression data showed that cross-linking with PEI and GPTMS greatly improved wet mechanical performance. The paper describes distinct compression regions, including an initial elastic region, a plateau associated with pore collapse, and densification at higher strain. The cyclic compression plots show that CGP3 had the strongest response of the tested beads.
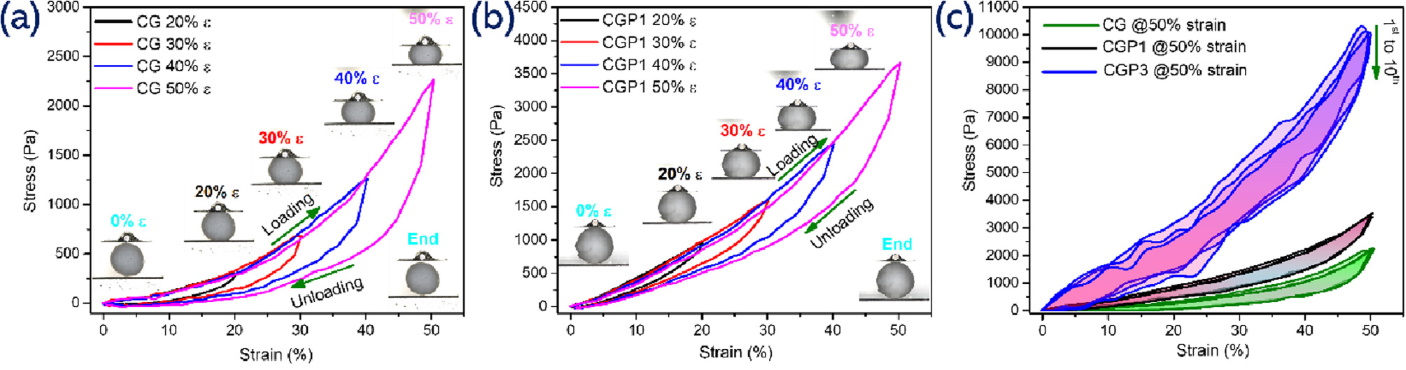
The numbers are especially useful. At 50% strain, CGP3 showed about 10.5 kPa stress and maintained very low permanent deformation, around 2.00%, even across repeated cycles. The same section also reports low energy loss coefficients for the cross-linked beads, consistent with good elasticity and recoverability under wet compression.
That makes the mechanical result more than a basic characterization step. It shows the beads were robust enough to survive the kinds of wet handling and loading conditions that matter for real adsorption use.
Copper adsorption performance
The adsorption results were strong. According to the Langmuir fits, the maximum Cu(II) adsorption capacities were 126.10 mg/g for CGP1 and 163.40 mg/g for CGP3. The higher-performing bead also had the higher amine content, which fits the proposed copper-chelation mechanism well.
The kinetics were also fast. The paper reports that equilibrium was reached in less than 10 hours, and it specifically notes that the smaller bead format improved adsorption by shortening diffusion distance and increasing available surface area. The kinetic analysis suggested pseudo-second-order behaviour and pore-diffusion involvement.
Why wet mechanical strength matters for adsorbents
Adsorption capacity is important, but a material also needs to survive immersion, handling, compression, and regeneration. A bead that adsorbs well but collapses in water is not especially useful. That is why the mechanical testing here adds so much value. It helps show that the beads are not just chemically active, but physically durable enough for reuse-oriented applications.
The paper even notes that the cross-linked structure made the beads promising for packed column adsorption, which is exactly the kind of application where wet robustness matters.
If you want to learn more about how the CellScale MicroTester supports mechanical testing of aerogels, beads, and porous biomaterials, visit the MicroTester product page.
Regeneration and reuse
A good wastewater adsorbent should not only remove contaminants once. It should also be recoverable and reusable. In this study, the absorbed copper could be removed using EDTA-Na2, and the paper reports no clear reduction in adsorption capacity over five regeneration cycles. That is a strong practical result, especially when combined with the mechanical stability data.
The study also tested the beads in simulated wastewater containing humic acid. Performance was slightly reduced compared with the simpler copper solution, but the beads still showed promising adsorption behaviour under those more realistic conditions.
Related reading
Read the full publication here: Shape recoverable and mechanically robust cellulose aerogel beads for efficient removal of copper ions.
For a related example of small-sample compression testing, see our post on pullulan microbead compression testing.
Final takeaway
This study is a good example of why mechanical characterization matters in materials-for-water-treatment research. The best result was not only that the beads removed copper effectively. It was that the same beads combined high adsorption capacity, fast uptake, wet mechanical robustness, and regeneration in one platform. For cellulose aerogel beads for copper removal, that is a much stronger story than chemistry alone.