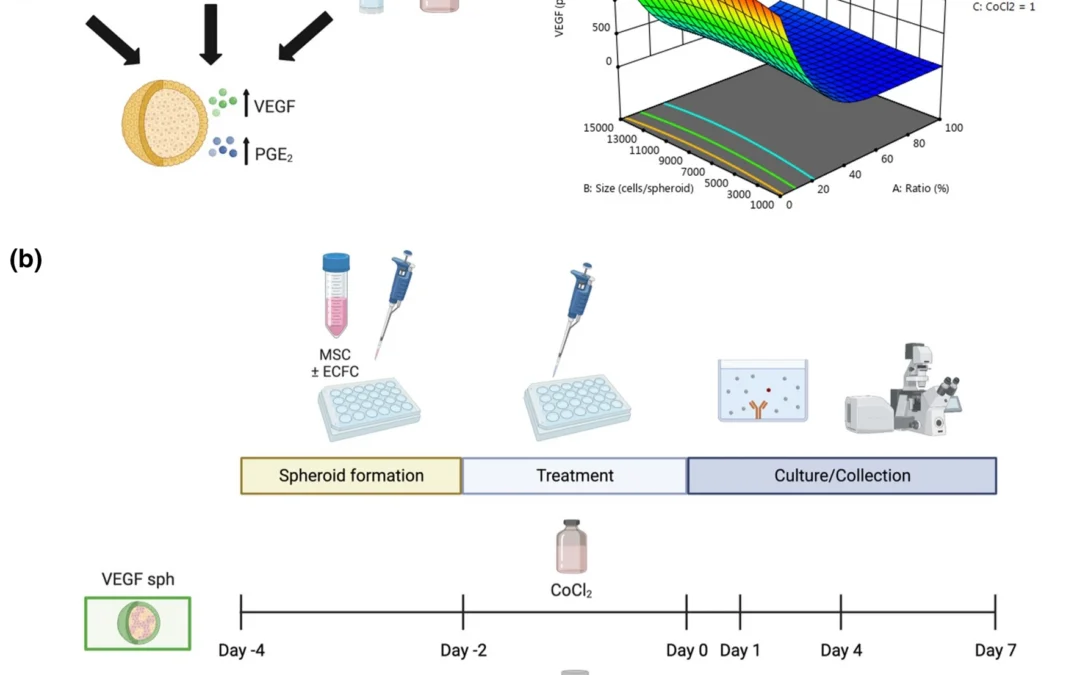
Mesenchymal stromal cell spheroids are already known to outperform dispersed cells in several regenerative settings, especially when cell survival and secreted repair signals matter. A study out of the University of California, Davis asked a more specific question: instead of making one spheroid type that does everything reasonably well, can you engineer different spheroids to do different jobs?
The researchers used a design of experiments approach to tune the spheroid microenvironment and generate two distinct formulations. One was optimized to maximize VEGF secretion and support angiogenic activity. The other was optimized to maximize PGE2 secretion and support epithelial repair. They also incorporated endothelial colony forming cells into the system to address a practical challenge in chronic wounds, where host endothelial cells may be dysfunctional and less responsive to pro-angiogenic cues.
Why mesenchymal stromal cell spheroids matter in wound healing
The appeal of mesenchymal stromal cell spheroids is not just that they group cells together. Aggregation changes how the cells behave. Compared with monodispersed MSCs, spheroids can show stronger survival in harsh environments and enhanced secretion of endogenous factors that matter for tissue repair, including VEGF and PGE2. The paper frames these as two particularly important signals in wound healing: VEGF for angiogenesis and PGE2 for inflammatory regulation and epithelialization.
What makes this paper useful is that it pushes beyond the usual “more is better” framing. The researchers wanted to separately tune the spheroids so they could use them in a more modular way.
How the researchers tuned the spheroid microenvironment
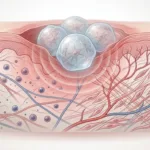
The study used a Box-Behnken design of experiments model to test how spheroid cell number, endothelial cell content, and biochemical treatment affected spheroid function.
For the VEGF-focused spheroids, the variables were:
-
total cell number
-
percentage of endothelial colony forming cells
-
CoCl2 concentration as a hypoxia-mimetic cue
For the PGE2-focused spheroids, the formulation was based on:
-
total cell number
-
endothelial cell percentage
-
Pam3CSK4 as an inflammatory stimulus
The optimized formulations were very different. The VEGFMAX spheroids used 8,000 cells, 0% ECFCs, and 95.4 μM CoCl2, while the PGE2,MAX spheroids used 60,000 cells, 33% ECFCs, and 1 μg/mL Pam3CSK4.
What the study found about spheroid function
The distinct formulations did exactly what the researchers hoped they would do.
The paper reports that:
-
VEGFMAX spheroids produced 22.7-fold more VEGF than PGE2,MAX spheroids in the abstract summary
-
PGE2,MAX spheroids produced 16.7-fold more PGE2 than VEGFMAX spheroids in the abstract summary
-
VEGFMAX spheroids drove stronger endothelial migration
-
PGE2,MAX spheroids drove faster keratinocyte migration
In the body of the paper, the validation data showed that VEGFMAX secreted much more normalized VEGF than the comparison spheroids, while PGE2,MAX produced the strongest PGE2 output. The two formulations also had different broader secretome profiles, with VEGFMAX dominated by VEGF-A, and PGE2,MAX showing much higher CD31, TIE-2, HGF, and EGF.
That is a more interesting result than a generic “improved wound healing potential” claim. These were not just better spheroids. They were different spheroids with different jobs.
Mechanical properties of the spheroids
Mechanical properties of the spheroids were evaluated at the microscale using a CellScale MicroTester to link physical cues with biological function. The authors measured spheroid diameter and elastic modulus after formation and found that the two optimized spheroid types were physically distinct as well as biologically distinct.
At 48 hours:
-
PGE2,MAX spheroids were much larger, about 750.9 ± 28.6 μm
-
VEGFMAX spheroids were smaller, about 323.6 ± 59.9 μm
-
VEGFMAX spheroids had the higher elastic modulus, about 3674 ± 1924 Pa
-
PGE2,MAX spheroids were softer, about 1741 ± 887 Pa
So this is not just a secretome story. The spheroids were mechanically different too.
Why microscale mechanics mattered here
The mechanical data help explain some of the later behaviour. The smaller VEGFMAX spheroids were more compact and mechanically stiffer, while the larger PGE2,MAX spheroids were softer. In the discussion, the authors connect those differences to cell spreading behaviour in hydrogels, suggesting that the more compact VEGFMAX spheroids may require more effort from cells to attach and spread into the surrounding matrix.
This is exactly why microtissue and spheroid mechanics matter. If two spheroids differ in size, compaction, and stiffness, they are likely to interact differently with a biomaterial carrier and with surrounding cells.
Bioactivity in endothelial and keratinocyte migration assays
The study did not stop with cytokine readouts. The authors tested the conditioned media from the two spheroid types in migration assays.
They found that:
-
VEGFMAX conditioned media promoted stronger migration of ECFCs
-
VEGFMAX media also improved migration of diabetic HMVECs, which is important because chronic wounds often involve dysfunctional endothelium
-
PGE2,MAX conditioned media promoted stronger migration of HaCaT keratinocytes
Those results reinforce the idea that the spheroids had been tuned into functionally distinct therapeutic tools rather than just different cell aggregates.
Behaviour in PEG-4MAL hydrogels
The final part of the paper moved the spheroids into MMP-degradable PEG-4MAL hydrogels as a model delivery system. Both spheroid types remained viable and spread into the material. The paper reports robust protrusion formation and matrix interaction over 7 days, with some evidence that combining the two spheroid types increased early protrusion number through paracrine interaction between them.
That makes the work relevant not only to wound healing, but also to:
-
organoid and tissue mimetic systems
-
stem cell mechanobiology
-
microtissue delivery in biomaterials
-
modular cell therapy design
Why this research matters
This paper is strong because it combines three things that are often treated separately:
-
microenvironmental tuning
-
biological function
-
physical characterization
The result is a clearer picture of how mesenchymal stromal cell spheroids can be engineered for different regenerative goals. It also gives the MicroTester a real role in the story, since the mechanical measurements helped show that the optimized spheroids were physically distinct, not just different in cytokine output.
Conclusion
This study shows that mesenchymal stromal cell spheroids can be tuned into functionally distinct microtissues rather than treated as one generic therapeutic format. By adjusting cell number, endothelial cell content, and biochemical cues, the researchers created one spheroid type that favored angiogenic signaling and another that favored epithelial repair. They also showed, using the CellScale MicroTester, that these spheroids differed mechanically at the microscale, with the VEGFMAX spheroids being smaller and stiffer than the larger, softer PGE2,MAX spheroids.
Read the full article here.