4D Biofabricated, Magnetically Augmented Callus Assembloids Accelerate Bone Formation
Recent advances at the intersection of mechanobiology and biomaterials engineering are reshaping how researchers approach regenerative medicine. A publication from early 2025 in Advanced Science demonstrates a large step forward for natural fracture healing research: a 4D biofabricated, magnetically responsive callus assembloid implant capable of dramatically accelerating endochondral ossification.
The researchers from Belgium, Canada, Spain, and the UK relied heavily on mechanical testing of biomaterials to determine how changes in stiffness, stress–strain behavior, and mechanical maturation influence mechanosensitive pathway activation. Using the CellScale MicroTester LT, they quantified how the assembloids stiffened over time and linked this mechanical evolution directly to activation of key mechanotransduction regulators such as YAP/TAZ and RUNX2.
This study not only shows how advanced biomaterials can guide bone regeneration, but also underscores why precise mechanical testing instrumentation is essential for validating next-generation engineered tissues.
Engineering a Mechanically Active, Magnetically Responsive Callus Assembloid
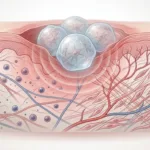
Central to the study is the callus assembloid, which is a 3D, multicellular construct designed to represent the early soft callus that forms during bone repair. This system undergoes 4D biofabrication, which means that its architecture and mechanical properties evolve over time.
In order to increase the assembloids regenerative capability, the researchers introduced magnetic nanoparticles (MNPs) into cartilaginous microtissues. These MNPs allow the constructs to be remotely manipulated during magnetically driven biofabrication, enabling:
-
Remote mechanical modulation
-
Architectural alignment via magnetic fields
-
Enhanced mechanotransduction
-
Tunable internal stiffness gradients
-
Improved organization of developing cartilage and bone tissue
This introduces a powerful framework for influencing mechanosensitive pathways critical to tissue maturation, particularly in bone repair, where local mechanical forces drive the structural progression from cartilage to mineralized bone.

Figure: Overview of the 4D biofabrication workflow. Cartilaginous microtissues are formed from hPDCs, loaded with magnetic nanoparticles, and assembled into callus assembloids using magnetically driven biofabrication. Post-fabrication stimulation further enhances maturation. Adapted from the original article (©2025 TheAuthor(s). Advanced Science published by Wiley-VCH GmbH).
Why Endochondral Ossification Is Mechanically Driven
Endochondral ossification is the body’s natural mechanism for repairing large bone defects. It involves:
- Formation of a soft cartilaginous callus
- Time-dependent increases in tissue stiffness
- Activation of mechanosensitive pathways
- Progressive mineralization and bone formation
This biology is inherently tied to biomaterial stiffness, mechanical loading, and cell–matrix mechanotransduction. Therefore, to engineer a successful implant, the authors needed precise data on how their constructs matured mechanically over time.
This is where mechanical testing of biomaterials was essential.
Magnetic Augmentation Activates Mechanosensitive Pathways
The study reports significant activation of mechanosensitivity-linked genes and transcriptional regulators, including:
-
YAP/TAZ nuclear localization, a hallmark of mechanotransduction
-
RUNX2, which promotes osteogenic differentiation
-
SOX9 and other chondrogenic markers involved in the cartilage-to-bone transition
Importantly, these biological changes aligned closely with the constructs’ mechanical maturation, reinforcing the connection between stiffness, mechanosensitive pathways, and accelerated bone formation (data quantified using the MicroTester LT).
Mechanical Testing of Biomaterials Using the CellScale MicroTester LT
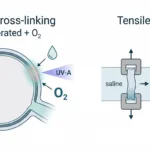
To establish the relationship between mechanical maturation and pathway activation, the researchers performed compressive modulus testing and stress–strain analysis using the CellScale MicroTester LT, a low-force mechanical testing instrument capable of force resolution down to 50nN (designed for hydrogels, soft tissues, micro-scale biomaterials, and other applications).
The mechanical testing of biomaterials was used to:
-
Measure changes in compressive modulus over multiple timepoints
-
Evaluate developmental stiffening consistent with chondrogenic → osteogenic transition
-
Compare magnetic versus nonmagnetic assembloids
-
Characterize nonlinear stress–strain behavior indicative of maturing bone-like tissue
-
Validate that magnetic augmentation leads to faster acquisition of load-bearing capacity
These data were central to proving that the construct’s enhanced stiffness drives mechanosensitive signaling and accelerates endochondral ossification.
Compression Response at 0% and 15% Strain

Figure 6g: Compressive deformation of magnetically augmented callus assembloids at Day 7. Images show the construct at 0% strain and at 15% strain during mechanical testing. Adapted from the original article (©2025 TheAuthor(s). Advanced Science published by Wiley-VCH GmbH).
Mechanical testing of biomaterials revealed a steeper rise in modulus for magnetically patterned assembloids. At Day 7, the constructs display visibly increased compaction at 15% strain compared to 0%, illustrating their early-stage mechanical behavior. This stiffening trend coincided with gene expression patterns tied to ossification, supporting the mechanobiology principle that increasing stiffness promotes osteogenic differentiation.
Stress–Strain Curves Demonstrate Accelerated Stiffening

Figure 6h: Stress–strain response of magnetically augmented (Mag+) and control assembloids at Days 7 and 21. Magnetically augmented constructs exhibit higher stiffness and improved load-bearing behavior as they mature. Adapted from the original article (©2025 TheAuthor(s). Advanced Science published by Wiley-VCH GmbH)
The mechanical signatures revealed higher peak stress values, more nonlinear elastic behavior, improved mechanical resilience, and accelerated conversion toward bone-like mechanical properties. Without performing mechanical testing of biomaterials, these conclusions would not be possible.
In Vivo Results: Faster, More Robust Bone Formation
When implanted, magnetically augmented assembloids demonstrated faster mineralization, more mature bone formation, greater tissue integration, and accelerated bridging of defects
Together, the mechanical testing results and biological data show that these constructs achieve both mechanical and functional improvements that translate into better regenerative outcomes.
Why This Study Matters
This work demonstrates that:
1. Mechanics are as important as biology
Mechanical properties (not just biochemical cues) drive ossification.
2. Mechanical testing of biomaterials is essential
The researchers needed precise modulus and stress–strain data to understand how stiffness controlled mechanotransduction.
3. Magnetic biomaterials broaden regenerative capabilities
Remote, non-invasive mechanical modulation opens new paths for controlled tissue development.
4. 4D biofabrication is a powerful platform
Time-evolving constructs allow researchers to engineer biological progression in ways previously impossible, using high-sensitivity testing tools like the CellScale MicroTester in advancing mechanobiology and tissue engineering research.
Explore the CellScale MicroTester
The MicroTester is designed specifically for precision mechanical testing of biomaterials, including hydrogels, soft tissues, engineered cartilage, and microscale constructs like the assembloid in this study.
Its sensitivity, modularity, and low-force measurement capabilities make it ideal for mechanobiology, soft tissue biomechanics, tissue engineering, organoid modeling, organ-on-a-chip, 3d bioprinting, spheroid mechanics, and other research.
Read the Full Open-Access Publication
(© 2025 The Authors, published under CC-BY 4.0)