Fast fabrication matters in organ-on-a-chip work, but speed alone is not enough. If a 3D printed microtube is going to stand in for something like a small blood vessel, kidney tubule, or cochlear duct, it also needs the right size, shape, elasticity, and resistance to deformation.
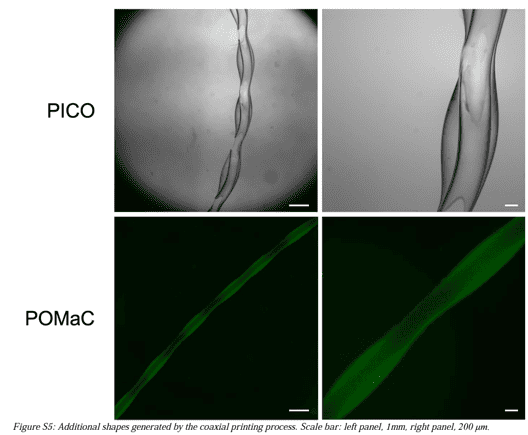
In a study published in Advanced Healthcare Materials, researchers at the University of Toronto developed a coaxial omnidirectional printing method for producing perfusable elastomeric microtubes from biodegradable polymers. The approach produced tubes with inner diameters of about 350 to 550 μm and wall thicknesses of about 40 to 60 μm, which is smaller than what their earlier coaxial printing methods could achieve. They also showed that the tube dimensions, stiffness, shape, and permeability could all be tuned in a controlled way.
Why 3D printed microtubes matter
Microscale tubular structures are everywhere in the body. Blood vessels, kidney tubules, and parts of the inner ear all depend on small, soft, mechanically functional tube-like geometries. The paper makes the point that these structures are not just conduits. Their dimensions and mechanical properties are tied to function. In vascular tissues especially, elasticity matters.
That has been one of the limitations of many hydrogel-based tube systems. Hydrogels can be useful because of their water content and cell-friendly nature, but they often deform too easily or collapse when cells remodel the surrounding matrix. The motivation here was to build something more mechanically robust using synthetic elastomeric polymers rather than relying only on conventional hydrogels.
What the researchers printed
The group used two photocrosslinkable elastomers:
-
PICO
-
POMaC
These were printed through a 21G/28G coaxial needle with the elastomer as the outer phase and Pluronic F127 as a fugitive inner phase. After printing into a Pluronic bath, the structures were photocrosslinked and the inner phase was removed to leave hollow, perfusable tubes.
The method was versatile enough to generate:
-
straight tubes
-
spiral tubes
-
tubes with wavy lumens
-
shapes resembling the cochlear duct
-
shapes resembling a kidney glomerular capillary
That is one of the more interesting parts of the paper. The method was not limited to straight channels. It could generate biomimetic tubular shapes that would be difficult to make quickly with older approaches.
Mechanical testing of the printed tubes
The researchers did not just print the tubes and show images. They mechanically tested them in two different ways.
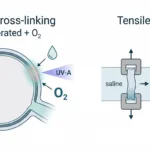
First, they used a CellScale BioTester for bulk uniaxial tensile testing of elastomer sheets made from PICO and POMaC, with alginate used as a hydrogel comparison. Those tests showed that the elastomeric materials were much stronger than the alginate control and behaved more like elastic materials than conventional hydrogel tubes.
Second, they used a CellScale MicroTester G2 to perform cyclic radial compression testing on the printed microtubes in aqueous solution at 37°C. The tubes were compressed to 10% strain over repeated cycles so the group could assess how much force was needed to deform them and how well they resisted radial collapse.
Why mechanical testing mattered here
The mechanics were central to the paper, not just an extra characterization step.
Mechanical testing of the 3D printed microtubes was essential for validating flexibility, radial stiffness, and deformation behaviour under physiologically relevant loading.
That is exactly what the study did. The authors were trying to make microtubes that could better match native vascular mechanics and avoid the collapse or irreversible deformation seen with softer hydrogel systems.
What the BioTester tensile testing showed
In the tensile work, the elastomer sheets were tested under cyclic uniaxial stretch. The paper reports that PICO and POMaC were both much stronger than 2 w/v% alginate. The tube demonstrations supported that result visually as well.
The alginate tubes could be stretched substantially, but they showed obvious permanent deformation after loading. The PICO and POMaC tubes, by contrast, behaved elastically and returned much more closely to their original geometry. The authors also compared these tubes with rat femoral vessel explants and found that the natural vessels showed behavior that fell between the responses of the two printed elastomer systems.
That comparison matters because it shows the researchers were not just chasing strength. They were trying to reach a useful mechanical range relative to native tissue.
What the MicroTester compression tests showed
The researchers compressed the tubes radially in water at 37°C using a plate attached to a microbeam and calculated stiffness from force-displacement response during repeated 10% strain cycles. They compared multiple PICO tube conditions generated through a design of experiments approach against alginate tubes and rat femoral vessels of similar outer diameter.
The result was clear:
-
all of the PICO tubes were much stiffer than the alginate tubes
-
the alginate tubes had very low stiffness and were not significantly different from zero in the reported comparison
-
the lower-stiffness PICO tube conditions fell into the same general range as the ex vivo rat vasculature
That is an important finding because it shows the 3D printed microtubes could be tuned toward a more tissue-relevant mechanical window rather than being either too floppy or unrealistically rigid.
Tunable dimensions and stiffness
The authors also used a design of experiments framework to tune the tube properties. They varied:
-
overall flow rate
-
inner-to-outer flow rate ratio
-
needle height above the support bath
-
needle speed
-
UV exposure energy
Using those factors, they were able to control both inner diameter and wall thickness, while also tuning radial stiffness. This is one of the more useful parts of the paper for people doing device development. The 3D printed microtube is not fixed to one geometry or one mechanical condition. The process can be adjusted to reach a target range.
Post-printing control over permeability
Another interesting feature of the study is that permeability was not treated as fixed. After printing, the researchers used femtosecond laser ablation to create microholes of about 10 to 20 μm in the tube walls. These holes increased permeability and were intended to help model exchange across vascular-like structures.
This matters because it expands the possible uses of the system. A 3D printed microtube can be mechanically robust and still be modified to allow diffusion or cell movement across the wall, which is valuable for organ-on-a-chip and tissue interface models.
High-throughput organ-on-a-chip fabrication
The paper also makes a practical manufacturing point. The tubes could be printed directly onto a 96-well plate-based organ-on-a-chip platform. The authors report that printing microhole-free tubes across an entire 96-well plate took about 3.6 minutes, whereas a comparable earlier manual stamping approach took 1.5 to 6.5 days depending on preparation steps.
That speed matters because a lot of promising organ-on-a-chip methods remain too slow or too manual to scale well for screening work. Here, the printing method was fast enough to support a more practical fabrication workflow.
Why this research matters
This paper is useful because it solves several problems at once. It improves the scale of elastomeric tube printing, gives better control over mechanical properties, allows post-printing permeability tuning, and connects directly to organ-on-a-chip applications.
It is also a strong example of how different test modes support different parts of the same development process:
-
BioTester for bulk tensile behaviour of elastomer materials
-
MicroTester for microscale radial compression of the printed tubes
That combination is what made it possible to show that the printed structures were not only small and fast to fabricate, but also mechanically meaningful.
Conclusion
This study shows that 3D printed microtubes can be made quickly without giving up mechanical control. Using coaxial omnidirectional printing, the researchers fabricated perfusable elastomeric microtubes with tunable size, radial stiffness, shape, and permeability. Mechanical testing with both the CellScale BioTester and CellScale MicroTester G2 helped show that the elastomeric tubes behaved much more like native soft tubular tissues than alginate-based hydrogel tubes and could be tuned into the same general mechanical range as ex vivo rat vasculature.
That makes the work relevant not only for organ-on-a-chip systems, but also for broader efforts in biofabrication, soft tubular biomaterials, and microphysiological model development.
You can read the full study here.
Related reading: For another example of how mechanical testing supports advanced 3D tissue models, read our post on the 3D Bioprinted Pterygium Eye Model for Disease Research and Drug Testing.