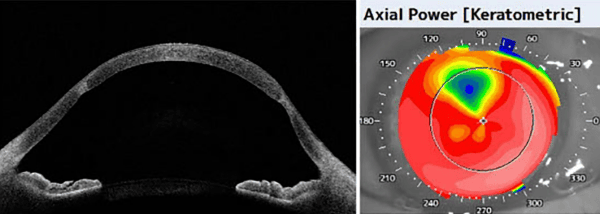
Most people think of corneal cross-linking as a treatment for keratoconus in the central cornea. This corneal stiffening study looked somewhere else. The group in Vienna wanted to know whether a similar idea could be used in the peripheral cornea to help the tissue around a graft stay mechanically stable over time after transplantation.
That view matters because the transition zone between donor and recipient tissue can become a site of recurrent keratoconic change over time. If the peripheral recipient bed continues to weaken, the graft may tilt or decenter, leading to high astigmatism and reduced vision. In this study, the researchers tested whether peripheral corneal cross-linking could increase biomechanical stability in the 9 to 11 mm corneal zone and potentially support better transplant outcomes.
Why peripheral corneal stiffening matters
The paper makes a practical clinical point early on. Standard cross-linking protocols are designed for the central cornea, but the periphery is thicker and mechanically different. That means a protocol that works centrally may not produce the same effect in the peripheral tissue.
The motivation here was not routine keratoconus treatment alone. It was surgical optimization. The authors were interested in whether stiffening the peripheral cornea before or during keratoplasty-related planning could improve topographic transplant stability and reduce the risk of recurrent ectatic change at the graft-host junction.
That makes this work relevant not only to keratoconus management, but also to:
-
corneal implant evaluation
-
ophthalmic surgical optimization studies
-
broader soft tissue biomechanics workflows in ophthalmology
What the researchers tested
The study used 36 human donor corneas for peripheral cross-linking experiments and varied two treatment parameters:
-
total energy dose: 5.4, 7.2, and 10 J/cm²
-
oxygen environment: 21% oxygen or 100% oxygen
Cross-linking was applied only to the peripheral 9 to 11 mm ring of the cornea, not the central treatment zone used in conventional protocols. Each cornea was divided so that one region was treated under normoxic conditions, one under hyperoxic conditions, and one left untreated as a control.
How biomechanical stiffening was measured
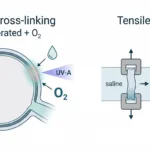
Biomechanical stiffening was quantified through controlled uniaxial mechanical testing of peripheral corneal tissue.
After treatment, the peripheral corneal ring was dissected and each section underwent stress-strain testing using a CellScale UStretch (superseded by the UniVert). Throughout testing, hydration was controlled in a 16% dextran bath. A prestress was applied, then strain was increased linearly, and the force required to reach 12% strain was recorded as the biomechanical endpoint.
What the study found
The main result is that standard-fluence peripheral treatment was not enough.
The paper reports that:
-
5.4 J/cm² at 21% oxygen did not produce a significant increase in rigidity in the peripheral cornea
-
increasing energy improved biomechanical response
-
adding supplemental oxygen further improved the effect in the higher-energy protocols
-
the most effective practical protocols were 7.2 J/cm² with 100% oxygen and 10 J/cm² without supplemental oxygen
That is the real takeaway. The peripheral cornea did respond to cross-linking, but it required a stronger or modified protocol than the standard central treatment approach.
Resistance to digestion supported the mechanical findings
The study did not rely on stress-strain testing alone. The authors also used collagenase A-assisted digestion to measure how long treated tissue resisted enzymatic breakdown.
Those results followed the same general trend as the mechanical testing:
-
higher fluence increased resistance to digestion
-
supplemental oxygen improved resistance further
-
the strongest effects were seen in the higher-energy groups, especially under hyperoxic conditions
That gives the paper more weight, because the mechanical stiffening data and the digestion resistance data pointed in the same direction.
What the histology showed
The histology is also important here. TUNEL assay and H&E staining showed that increasing energy increased the depth and extent of stromal cellular effect.
In the lower-energy protocol, the effect remained relatively superficial. At 7.2 J/cm², the treatment effect became more substantial, though still not as deep as the highest setting. At 10 J/cm², especially with supplemental oxygen, the effect extended much deeper into the stroma.
That deeper effect may be useful if the goal is stronger peripheral stabilization, but it also introduces a safety question. The authors specifically note that high-fluence treatment with supplemental oxygen raised concern about endothelial safety, even though the best protocols overall were still identified as 7.2 J/cm² with 100% oxygen or 10 J/cm² without supplemental oxygen.
Why this matters for surgical outcomes
The value of corneal stiffening in this study was not just to stiffen tissue in general. It was to potentially improve the stability of the recipient peripheral cornea in cases where long-term graft shape can be affected by progressive biomechanical weakness.
That makes the study relevant to:
-
keratoplasty planning
-
corneal transplant stability
-
recurrent keratoconus risk
-
corneal implant evaluation
-
ophthalmic device and procedure optimization
It is a good example of how mechanical testing can support a surgical question rather than only a materials question.
Conclusion
This study showed that peripheral corneal stiffening can be achieved, but not reliably with the standard central corneal cross-linking protocol alone. In the peripheral 9 to 11 mm zone, stronger or modified protocols were needed to produce a meaningful biomechanical effect. Stress-strain testing with a CellScale UStretch showed that 7.2 J/cm² with 100% oxygen and 10 J/cm² without supplemental oxygen were the most effective treatment conditions among those studied.
Read the full publication here.