Melt processing is widely used to shape thermoplastics in industry, but it is not always easy to bring into academic biomaterials research. Standard systems are often expensive, require large material volumes, and leave too much dead volume for small-batch experimental work. That is what makes this low-cost pilot-scale melt processing system so useful. It was designed specifically to lower the barrier to pilot-scale fabrication for academic labs working with limited amounts of polymer and composite materials.
The publication out of UC San Diego describes a benchtop extrusion and injection-molding device built from repurposed 3D-printer parts and a pneumatically driven piston. The system was designed for polymer processing at scales below 0.02 cm3, using total sample masses in the 50 to 500 mg range. That is a much better fit for many research labs than conventional commercial systems that may require tens to hundreds of grams of material just to run a single test.
One reason this matters for biomaterials is that melt-processed polymers are already used in tissue engineering scaffolds, microneedles, microfluidic devices, and controlled drug-delivery systems. A smaller, lower-cost platform makes it easier to explore those kinds of applications earlier in development.
For a broader application area, see our page on Polymers & Elastomers Testing.
Why pilot-scale melt processing is hard in academic labs
The paper makes the problem clear. Melt processing is highly scalable, solvent-free, and common in industrial thermoplastic manufacturing, but academic researchers often avoid it because the equipment is expensive and the required material volumes are too large for high-value experimental formulations. That is especially true when the materials contain pharmaceuticals, proteins, nanoparticles, or specialty fillers that are costly to make in bulk.
That is what this low-cost pilot-scale melt processing system was designed to address. Instead of replicating a full industrial machine, the authors created a compact test-bed for early-stage formulation screening and pilot studies.
How the melt processing system was built
The device design centered on a fused deposition modeling 3D-printer heater block mounted on T-slot aluminum rails, with a pneumatic cylinder used to drive a plunger through a heated barrel. The paper explains that this piston-based approach was chosen for simplicity, low cost, and low dead volume.
The materials cost was kept very low. In the conclusion, the authors report that the machine itself could be built for about $300, with custom molds costing roughly $200 per mold, which is far below the cost of traditional injection-molding tooling. That is a big part of the appeal for academic labs.
Injection molding small polymer composite batches
To demonstrate the system, the authors processed PLA, PLGA, and PCL along with several dispersed fillers, including nickel-coated multiwall carbon nanotubes, ferrocene, and doxorubicin. The processing workflow involved a first extrusion step to homogenize the materials, followed by injection into custom CNC-machined aluminum molds.
This is where the paper becomes especially practical. It does not just present a concept. It shows a workable route for injection molding for biomaterials using small sample masses and accessible hardware.
What the materials characterization showed
After molding, the polymer composites were characterized by SEM and EDS to evaluate morphology and filler distribution. The results showed that solvent-free PCL and PLGA samples had fewer voids than PLA samples prepared through solvent-based steps. The electron micrographs in the study show this clearly, and the discussion ties the lower porosity in the solvent-free samples to the absence of trapped solvent and improved powder pre-compression during loading.
The EDS results were especially useful for the nanotube- and ferrocene-loaded samples. In the solvent-free PCL samples, additive distribution was much more uniform and aggregation was lower than in the solvent-processed PLA samples. That gave the authors a simple way to compare how processing route affected dispersion quality.
Why tensile testing mattered in this study
The paper did not stop at fabrication and microscopy. It also used tensile testing of polymer composites to check how processing conditions affected mechanical performance.
For the tensile tests, the team injection molded reduced-size dog-bone specimens based on ASTM concepts, then tested them on a CellScale UniVert with a 100 N load cell.
That makes this a good example of mechanical testing of injection molded biomaterials. The UniVert data were used to assess whether the pilot-scale processing method produced mechanically reasonable parts and how solvent versus solvent-free preparation affected the final material response.
For more on the method side, see our page on Tensile Testing.
What the mechanical testing showed
The mechanical results were one of the clearest takeaways in the paper. For solvent-free PLA composites, tensile strength was largely independent of the injection temperatures and pressures tested. In contrast, the solvent-processed PLA samples showed much lower strength, and that drop became worse at higher injection temperatures.
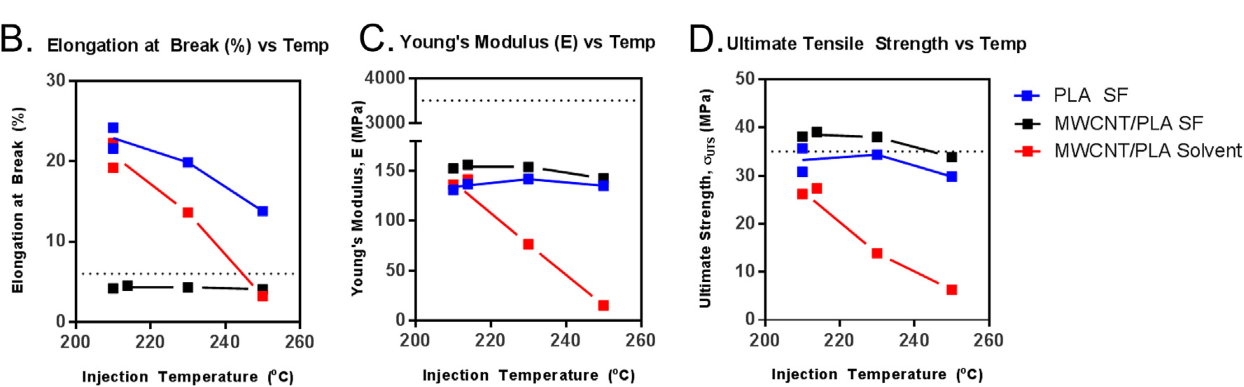
The paper also reports that the solvent-free samples were much closer to literature values for pristine PLA. They found a mean density of 1.236 g/cm3, ultimate tensile strength of 37.3 MPa, and elongation at break of 4.3% for solvent-free PLA+MWCNT, compared with much lower strength for the solvent-processed group. The conclusion was that solvent-free preparation preserved mechanical performance much better.
That is a useful result for labs interested in tensile testing of polymer composites after small-batch processing. It shows that the benchtop system was not only convenient, but capable of producing parts with meaningful mechanical properties.
If you want to learn more about the system used for those tests, visit the UniVert product page.
Shear rate and biologically sensitive materials
One especially helpful part of the paper is the discussion of shear. Because some biomedical payloads are sensitive to heat and shear, the authors modelled the theoretical shear rates inside the nozzle using a non-Newtonian COMSOL approach. The simulations in the paper show how shear rate changes with barrel pressure, barrel diameter, and nozzle size. The discussion explains that shear rate depends mainly on nozzle size and barrel pressure, and can be reduced by lowering extrusion speed.
That matters because this is not only a plastics paper. It is also relevant to biomaterials manufacturing, where proteins, pharmaceuticals, or nanoparticles may not tolerate aggressive processing conditions.
Why this matters for biomaterials and medical devices
The paper repeatedly ties melt processing back to biomedical use. In the introduction, the authors note that melt-processing has already been used for tissue engineering scaffolds, microneedles, microfluidic devices, and controlled release systems. They also point out that melt processing avoids solvents and may reduce microbial contamination because of the heat and pressure involved.
That makes this a good research-to-manufacturing highlight. It shows how a fabrication system can be scaled down enough for academic use without losing the connection to real polymer-processing workflows used in biomedical device development.
Where CellScale fit into the study
The CellScale UniVert was used to quantify the mechanical properties of the molded polymer composites, including elongation at break, Young’s modulus, and ultimate tensile strength. That was important because it gave the authors a direct way to compare the consequences of different processing approaches.
Without that tensile testing step, the study would still have shown that the machine could make parts. With it, the study showed something more useful: the system could make parts whose mechanical performance could be assessed in a meaningful way. That is a much stronger validation of a pilot-scale fabrication platform.
Related reading
The full paper can be read here: Design and fabrication of a low-cost pilot-scale melt-processing system.
If you are interested in the effects of cyclic and shear stretch on inflammation and tissue formation, go here.
Final takeaway
This study is best understood as a practical bridge between lab-scale formulation work and scalable polymer manufacturing. The low-cost pilot-scale melt processing system gave researchers a way to perform low-volume extrusion and injection molding with limited material, while still checking filler dispersion and using tensile testing of polymer composites to confirm mechanical performance. For academic biomaterials labs, that is a meaningful capability.