Engineering articular cartilage is difficult, but not because cells are unable to produce extracellular matrix. More often, problems show up later, when the tissue that forms lacks the internal organization needed to behave like cartilage. In native tissue, collagen fibers are arranged in a depth-dependent manner, running parallel to the surface near the top and reorienting at depth. That arrangement strongly influences how cartilage carries tensile load, but it has proven difficult to recreate once the scaffold is removed.
In cell-only bioprinting of articular cartilage, many of the cues that normally help guide matrix organization are deliberately absent. Cells are placed at high density and left to organize their own environment, which often results in tissue that is compositionally cartilage-like but structurally simplified. In a recent study published in Bioactive Materials, researchers from Trinity College Dublin and the Royal College of Surgeons in Ireland examined whether physical boundaries alone could bias how collagen alignment develops under these conditions.
Rather than introducing new materials or biochemical signals, they focused on limiting the space available to the tissue and then asking, through mechanical testing, whether those structural differences altered how the constructs deform under load. To do the mechanical testing, bioprinted cartilage sheets underwent tensile testing using a CellScale BioTester.

(A) Conceptual illustration showing how physical boundary spacing influences collagen alignment in scaffold-free cartilage constructs. Narrower confinement biases collagen organization along the long axis of the tissue, while embedded support baths (B) can act as temporary physical boundaries during cell-only bioprinting. Image adapted from Karam et al., Bioactive Materials (2026), CC BY 4.0.
Why cell-only bioprinting of articular cartilage struggles with structural organization
In cartilage, organization is not just a structural feature but a mechanical one, influencing how tensile loads are distributed across the tissue thickness. Many bioprinting strategies assume that if cells are placed in the correct shape, structure will eventually follow. For cartilage, that assumption often falls apart. Collagen alignment in scaffold-free constructs tends to be weak unless something actively biases how cells organize their surrounding matrix.
Rather than treating this as a materials problem, the authors frame geometry itself as a design variable. They ask how much structure might emerge if cells are simply prevented from expanding freely, and whether precisely defined physical boundaries can guide collagen organization during early tissue formation. This shift in perspective places physical confinement and boundary effects at the center of anisotropic cartilage tissue engineering.
Selecting a cell population that responds to physical confinement
Before introducing any form of confinement, bioprinting, or mechanical testing, the authors first considered which cells were most likely to respond to physical cues rather than ignore them.
Why articular cartilage progenitor cells were used
Articular cartilage progenitor cells are often described as more adaptable than mature chondrocytes, particularly during early matrix deposition. In pellet culture, these cells produced higher levels of cartilage-associated matrix than donor-matched chondrocytes while maintaining a stable phenotype (see Rikkers et al., 2021). Rather than treating this as a defining biological result, the authors use it pragmatically. Progenitor cells have been shown to reorganize and deposit extracellular matrix in a manner that is sensitive to their physical environment, making them a practical choice for studying how confinement and boundary conditions influence tissue organization.
Using physical confinement to guide collagen alignment in engineered cartilage
To explore how geometry influences matrix organization, the authors began their cell-only bioprinting of articular cartilage with a simple system: casting high-density, cell-only constructs inside agarose channels with defined widths.
Casting cell-only constructs within agarose boundaries
Agarose channels provided external physical boundaries that constrained lateral expansion while allowing cells to remodel their own matrix. Channels with widths ranging from 250 to 750 micrometers were used, with the same cell density applied across conditions. Over time, the constructs contracted, then expanded to fill the available space, forming handleable tissue filaments.

Cell-dense suspensions were cast into agarose channels with widths of 250, 500, or 750 µm, creating external physical boundaries that constrained lateral expansion during culture. After casting, constructs were crosslinked and maintained in chondrogenic media, allowing tissue formation under controlled geometric confinement.
Reproduced from the original publication under CC BY 4.0.
What narrower spaces appear to do to collagen organization
As boundary spacing decreased, collagen organization became more pronounced. Scanning electron microscopy showed collagen fibrils tending to align along the long axis of the narrowest channels, while wider channels produced more randomly oriented networks. Polarized light microscopy reinforced this observation, with higher fiber coherency near the boundaries in thinner channels.
Rather than presenting this as a binary outcome, the authors describe a gradual shift. Collagen alignment did not abruptly appear below a specific width, but instead became more consistent as confinement increased. This suggests that physical boundaries act less like templates and more like persistent biases that influence how tissue self-organizes.

(A) Scanning electron microscopy images showing increasing collagen fibril alignment as agarose channel width decreases. (B) Polarized light microscopy and corresponding colour maps show higher collagen coherency in constructs formed within narrower boundaries, indicating progressively aligned matrix organization along the construct axis.
Reproduced from the original publication under CC BY 4.0.
Cell-only bioprinting of articular cartilage using embedded support baths
Having established that external boundaries influence collagen alignment in cast constructs, the authors next asked whether the same principle could be translated into a bioprinting workflow.
Embedded support baths as temporary physical boundaries
To do this, they used an embedded bioprinting approach, extruding dense, cell-only filaments into a xanthan gum–based support bath. The bath provided temporary confinement, holding filaments in place during culture without acting as a permanent scaffold. This approach aligns with broader trends in embedded bioprinting of human tissue, where support baths are increasingly used to define geometry rather than deliver bioactivity.

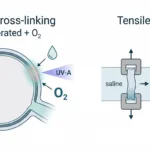
Articular cartilage progenitor (ACP) bioink is extruded into a methacrylated xanthan gum support bath, which behaves as a self-healing, deformable medium during printing. Adjusting print speed controls filament width and spacing, allowing internal boundaries or continuous filaments to be formed without gaps. Printed constructs are stabilized by brief UV crosslinking and maintained in chondrogenic culture for subsequent matrix development and structural organization.
Reproduced from the original publication under CC BY 4.0.
Filament width as a proxy for boundary spacing
By adjusting print speed, the authors produced filaments with different diameters while keeping cell density constant. As in the casting experiments, thinner filaments exhibited greater collagen alignment along their long axis. Polarized light microscopy showed higher coherency in narrow filaments, suggesting that confinement during culture, rather than shear during printing, played a dominant role.
Building anisotropic cartilage tissue through layered cell-only bioprinting
The next step was to move beyond single filaments and assemble sheet-like constructs that more closely resemble cartilage tissue.
Assembling anisotropic sheets from oriented filaments
The authors bioprinted sheets composed of orthogonally oriented filaments, creating regions with different principal directions. Cell viability remained high, and the constructs developed a cartilage-like matrix over time.

(A) Schematic illustrating the fabrication of anisotropic articular cartilage sheets by layering oriented cell-only filaments within a support bath, followed by UV crosslinking and chondrogenic culture. Distinct superficial and middle/deep zones are defined by filament orientation.
(B) Construct design and macroscopic appearance over culture time (day 0 to day 28), including top and side views and live/dead staining, demonstrating structural integrity and high cell viability.
Reproduced from the original publication under CC BY 4.0.
Depth-dependent collagen alignment begins to emerge
Polarized light microscopy revealed differences in collagen orientation between regions, with fibers aligned parallel to filament direction. While the resulting architecture did not fully recapitulate native cartilage, it began to show depth-dependent organization reminiscent of anisotropic cartilage tissue engineering strategies.
How internal physical boundaries influence tissue formation and organization
In a final set of experiments prior to mechanical testing, the authors introduced internal boundaries between adjacent filaments within multilayer constructs.

(A) Polarized light microscopy reveals that introducing internal physical boundaries during bioprinting promotes an arcade-like collagen architecture, with distinct alignment patterns in the superficial and middle/deep regions of the construct. (B) Orientation intensity profiles quantify these differences, showing preferential collagen alignment near the surface compared to deeper zones.
Reproduced from the original publication under CC BY 4.0.
Comparing constructs with and without internal constraints
Constructs with internal spacing showed more uniform matrix deposition throughout their thickness, while those printed without internal boundaries exhibited poor tissue formation in the core. The presence of boundaries appeared to influence not only collagen alignment, but also overall tissue development.
Polarized light microscopy revealed that constructs with internal boundaries developed collagen networks that more closely resembled arcade-like organization, with horizontally aligned fibers near the surface and vertical alignment deeper within the tissue.
Probing articular cartilage mechanics after cell-only bioprinting
After extended culture, the authors subjected bioprinted sheets to tensile testing to examine how these structurally organized tissues deform under load. The full-field strain maps below are particularly informative, revealing how deformation is distributed across the sheet rather than reducing mechanical behaviour to a single bulk modulus.

Uniaxial tensile testing of bioprinted articular cartilage progenitor (ACP) sheets was performed using a CellScale BioTester. The figure shows macroscopic views and full-field strain maps under unloaded and loaded conditions, illustrating how deformation develops across the construct during tensile loading and enabling calculation of ramp and equilibrium tensile moduli.
Reproduced from the original publication under CC BY 4.0.
Why tensile testing was necessary in this study
In this study of cell-only bioprinting of articular cartilage, tensile testing using a CellScale BioTester provided a way to probe whether the structural organization observed by microscopy translated into measurable differences in how the tissue deforms under load. Because the constructs were hydrated, low-stiffness, and still early in maturation, conventional mechanical testers would have been poorly suited to this task.
Related work from the same group has approached this question from a different angle, using tensile testing with the BioTester to examine scaffold-free cartilage grafts formed through microtissue fusion. In that study, mechanical measurements were used less as a benchmark and more as a way to interpret how tissue organization and density evolve as individual units merge into a larger construct.
Read alongside the present work, it becomes clear that mechanical testing is being used as a lens to think about organization, rather than as a final proof of function.
What physical boundaries may offer cell-only bioprinting of articular cartilage
The authors do not claim to have solved cartilage regeneration. The engineered tissues do not fully replicate native architecture, and mechanical testing remained limited to early time points. What it does offer is a clear demonstration that physical boundaries, even simple ones, can influence how scaffold-free cartilage organize during cell-only bioprinting of articular cartilage.
By treating confinement as a design input rather than a limitation, the authors highlight an approach that sits somewhere between free self-assembly and rigid scaffolding. In the context of cell-only bioprinting of articular cartilage, geometry and boundary conditions may prove to be as influential as materials or growth factors, particularly during early tissue formation.
For researchers working at the intersection of bioprinting, mechanics, and cartilage tissue engineering, this work suggests that paying closer attention to space, constraint, and deformation may help bridge the gap between matrix production and functional organization.
Read the Full Publication
About the CellScale BioTester
The CellScale BioTester is used for biaxial, tensile, compression, peel, fatigue, viscoelastic, and other mechanical testing methods of biomaterials, with up to 200N force capacity and digital image correlation capabilities. It is commonly applied in studies where specimens are fragile, highly compliant, or still undergoing biological maturation, including hydrogels, scaffold-free tissues, and engineered constructs.
In this study, the BioTester was used to perform uniaxial tensile testing on bioprinted articular cartilage progenitor cell sheets following in vitro culture. Testing was carried out in a hydrated environment, allowing ramp and equilibrium tensile moduli to be measured and full-field strain maps to be generated. These measurements provided a mechanical context for the structural organization observed by microscopy, helping relate collagen alignment and boundary-driven architecture to how the constructs deform under load.