Human induced pluripotent stem cell-derived cardiomyocytes are used all over cardiac tissue engineering and drug screening, but the same issue keeps showing up. Even when differentiation is consistent, the cells usually stop short of an adult-like state. That gap matters because immature cardiomyocytes do not reproduce adult physiology well enough for truly predictive readouts.
In this Nature Communications study out of Osaka University in Japan, researchers developed a simple 3D tissue-ring platform that promoted the mechanical maturation of stem cell-derived cardiomyocytes through spontaneously circulating re-entrant waves.
Mechanical force measurement is important in this context because contraction is one of the defining functional outputs of cardiomyocytes. A maturation strategy is more convincing when it improves not only gene expression and structural alignment, but also the force the tissue can generate. That is one reason this paper stands out. It combined tissue formation, pacing-like behaviour, and contractile force measurement of engineered cardiac tissues in the same overall system.
For readers interested in the broader application area, see our page on Cardiac Tissue Engineering & Mechanics.
How the self-organized tissue rings worked
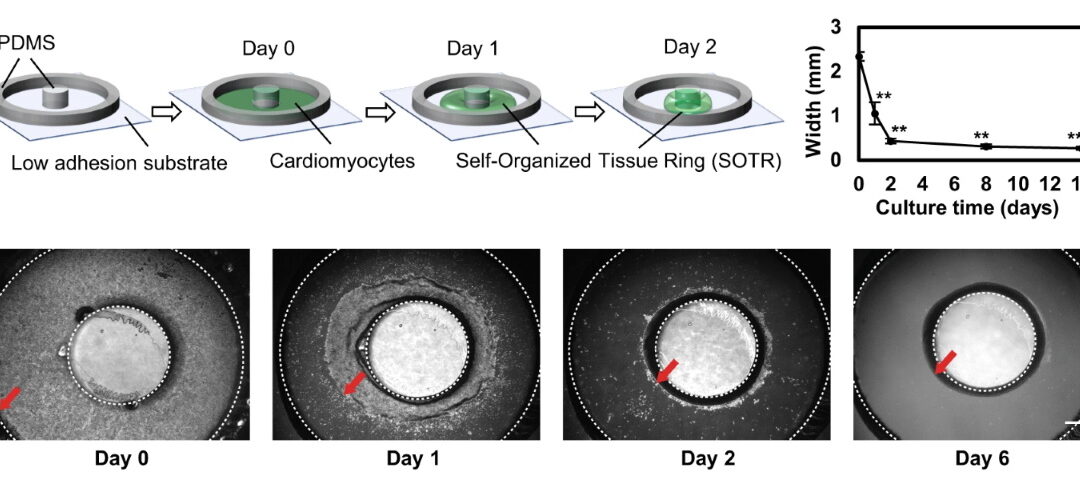
The authors created circular self-organized tissue rings, or SOTRs, by plating hiPSC-derived cardiomyocytes in a culture dish with a central PDMS pillar. Within two days, the cells aggregated around the pillar and formed ring-shaped tissues.
A key feature of the model was that re-entrant waves could arise spontaneously and travel around the closed loop. These waves made the tissues beat rapidly without requiring continuous external electrical stimulation. According to the paper, SOTRs with one or two re-entrant waves reached beating frequencies around 2.4 to 3.3 Hz on day 6, compared with only about 0.21 Hz in rings without re-entrant waves.
Why re-entrant waves mattered for cardiomyocyte maturation
The premise is simple: higher, sustained beating rates tend to push hiPSC-derived cardiomyocytes toward a more mature phenotype, but long runs of external pacing are not always clean or convenient. The re-entrant-wave setup takes a different route by generating rapid, persistent activation inside the ring itself. In the paper’s framing, this avoids some of the practical downsides that can come with high-frequency electrical stimulation over time, including electrode-driven chemistry changes and other culture-side artifacts.
That made the platform more than a tissue-formation method. It became a model for mechanical maturation of stem cell-derived cardiomyocytes driven by spontaneous internal pacing.
Structural and molecular signs of improved maturation
The maturation effects were broad. RNA sequencing and qPCR showed that SOTRs with re-entrant waves had higher expression of cardiac-related genes, including MYH7, ACTN2, DES, MYL2, MYL3, PLN, MB, and ADRB1. The PCA and clustering data show the re-entrant-wave groups separating from the zero-wave group, while the bar plots show higher expression of several maturation-related genes.
The structural data supported that result. The confocal images show that cardiomyocytes in the re-entrant-wave groups were more aligned along the ring direction, with stronger sarcomeric organization. Sarcomere length also increased, from about 1.49 µm in the zero-wave group to 1.71 µm with one re-entrant wave and 1.83 µm with two re-entrant waves. The TEM images further showed larger sarcomeric bundles and more defined ultrastructure in the trained tissues.
Improved Ca2+ handling and mitochondrial function
The functional gains were not limited to morphology and gene expression. The authors also examined mitochondrial respiration and Ca2+-handling. The two-wave group showed the highest maximal respiration capacity, indicating stronger mitochondrial activity. The same figure shows that caffeine-induced Ca2+ responses were also highest in the two-wave group, which the authors interpret as improved maturation of SR-related Ca2+-handling properties.
That combination is important because mature cardiomyocytes need both organized contractile structure and better calcium cycling to behave more like adult heart cells.
What the contractile force measurements showed
The authors measured contractility using a micron-scale mechanical tester from CellScale, the MicroTester benchtop system (formerly called the MicroSquisher). The SOTRs were removed from the pillar, cut into strips, fixed on a stage, immersed in warmed culture medium, and stretched with a cantilever beam so force could be recorded by beam deflection. The detailed setup is described in the contractility-analysis methods in the publication.
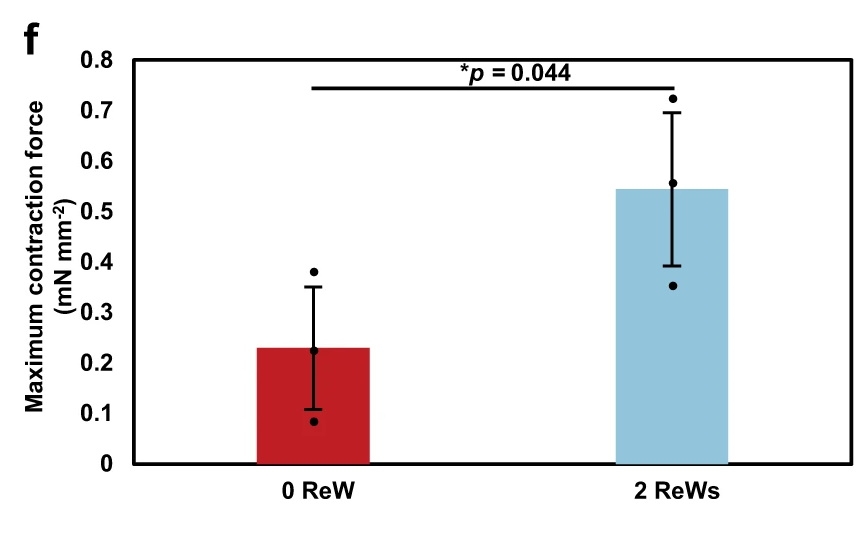
The result was clear: tissues with two re-entrant waves generated much stronger contractions than tissues with none. The paper reports maximum contractile force of about 0.54 ± 0.15 mN mm⁻² in the two-wave group versus 0.23 ± 0.12 mN mm⁻² in the zero-wave group. Both groups also showed a Frank-Starling-like increase in active force with greater stretch, but the re-entrant-wave-trained tissues produced larger amplitudes overall.
That is what makes this a strong example of contractile force measurement of engineered cardiac tissues. The MicroTester data did not just add a mechanical datapoint. They showed that the maturation strategy produced a meaningful functional gain.
For more on the method side, see our page on Micro-Mechanical Testing.
Why this matters for engineered cardiac tissues
A common challenge in cardiac tissue engineering is that engineered tissues may look organized without yet performing like more mature myocardium. This study addressed that directly. The re-entrant-wave-trained rings showed stronger organization, more mature gene expression, better Ca2+ handling, increased respiration, and greater force generation. Taken together, those outcomes make the system useful not only for tissue engineering, but also for drug screening and disease modelling.
For readers interested in adjacent model systems, see our page on Organoid and Tissue Mimetic Systems.
Where the MicroTester fit into the study
The CellScale role here was not peripheral. The MicroTester-based force assay was one of the most important functional readouts in the paper. It helped show that the observed maturation was not only molecular or structural. It also changed the mechanical performance of the tissue.
That is an important distinction. In a cardiac model, function matters. The ability to measure force in a controlled, small-scale setup made it possible to show that the re-entrant-wave condition improved the contractile behaviour of the engineered tissue itself.
Related reading
The full paper can be read here: Circulating re-entrant waves promote maturation of hiPSC-derived cardiomyocytes in self-organized tissue ring.
If you want another example of CellScale testing applied to engineered tissues, you may also be interested in compressive testing of rat and pig optic nerve head.
Final takeaway
This study is best framed around mechanical maturation of stem cell-derived cardiomyocytes. The self-organized tissue rings did not simply beat faster. They developed stronger cardiac organization, more mature molecular features, improved Ca2+ handling, and higher contractile force. By adding contractile force measurement of engineered cardiac tissues, the paper showed that re-entrant-wave training improved function as well as appearance.